PHYTOCHEMICAL PROFILING, ANTIOXIDANT, ANTITYROSINASE AND ANTIPLASMODIAL ACTIVITIES OF CHLOROFORM AND N-BUTANOL STEM FRACTIONS OF PARQUETINA NIGRESCENS (AFZEL.) BULLOCK: A COMPARATIVE STUDY
DOI:
https://doi.org/10.33003/fjs-2026-1002-4651Keywords:
Parquetina nigrescens, GC–MS, antioxidant activity, tyrosinase inhibition, antiplasmodial activity, selectivity indexAbstract
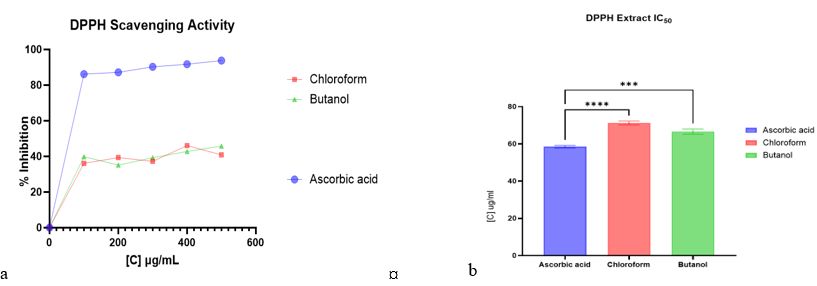
The therapeutic potential of the solvent partitioned fractions of Parquetina nigrescens (Afzel.) Bullock was evaluated using an integrated approach that included phytochemical screening, GC-MS analysis, antioxidant, antityrosinase, antimalarial, and cytotoxicity assays. The methanolic extract of the stem was subjected to successive partitioning using chloroform and n-butanol solvents. The phytochemical screening of the chloroform and n-butanol fractions showed the presence of alkaloids, flavonoids, phenolic, and terpenoid compounds, whereas the n-butanol fraction was predominantly rich in tannins and saponins. The GC-MS analysis identified 33 and 8 compounds from the chloroform and n-butanol fractions, respectively, which included fatty acid methyl esters, diterpenoids, and hydrocarbons. The fractions demonstrated moderate DPPH radical scavenging activity (IC50: 71.25 ± 1.05 µg/mL for chloroform fractions; 66.59 ± 1.40 µg/mL for n-butanol fractions) and nitric oxide scavenging activity (IC50: 71.45 ± 0.86 µg/mL for chloroform fractions; 73.17 ± 1.42 µg/mL for n-butanol fractions). The n-butanol fraction exhibited strong tyrosinase inhibition (IC50: 54.39 ± 1.30 µg/mL), surpassing kojic acid. Both fractions showed significant activity against Plasmodium falciparum (IC50 range: 3.224–3.611 µg/mL) with cytotoxicity IC50 values above 30 µg/mL on HeLa cells, yielding selectivity indices greater than 10. The pharmacological relevance of the fractions and the bioassay results validate the utility of the solvent partition method for the fractionation of P. nigrescens. The results also show the potential of the chloroform and n-butanol fractions as lead compounds for the development of antimalarial and tyrosinase inhibitors.
References
Adase, E., Ankutse, P., Kumadoh, D., Archer, M. A., Kyene, M. O., Yeboah, G. N., & Asamoah Agyare, D. O. (2022). A Review Of Parquetina nigrescens (Afzel.) Bullock, A Plant For Traditional Medicine: Phytochemical And Pharmacological Properties. Evidence-Based Complementary And Alternative Medicine, 2022, 6076707. Https://Doi.Org/10.1155/2022/6076707
Adekilekun, A. H., Omodamiro, O. D., Achi, N. K., Oyewusi, H. A., Omodamiro, R. M., Tijani, B. A.,. & Oyeyemi, B. F. (2025). Evaluation of Bioactive Compounds, In-Vitro Antioxidant Profile And Anti-Inflammatory Properties of Ethanolic Extracts of Isoberlinia Tomentosa. Fudma Journal of Sciences, 9(12), 148-158.
Adoga, S., Ekle, D., Kyenge, B., Aondo, T., & Ikese, C. (2019). Phytochemical screening, thin-layer chromatography and antimicrobial activity study of Parquetina nigrescens leaf extracts. Ovidius University Annals of Chemistry, 30, 88–94. https://doi.org/10.2478/auoc-2019-0016
Ajayi, L., Ayeleso, A., Oyedepo, T., & Mukwevho, E. (2021). Ameliorative potential of hydroethanolic leaf extract of Parquetina nigrescens on D-galactose-induced testicular injury. Molecules, 26(11), 3424.
Apalla, Z., Nashan, D., Weller, R. B., & Castellsagué, X. (2017). Skin Cancer: Epidemiology, Disease Burden, Pathophysiology, Diagnosis, and Therapeutic Approaches. Dermato-Endocrinology, 9(1), e1269035. Https://Doi.Org/10.1080/19381980.2016.1269035
Ayoola, M., Ogundeko, Y., Obanleowo, T., Omole, D., Chukwu, B., & Faloye, K. (2024). Evaluation of the antidiabetic activities of the fruit of Parquetina nigrescens (Afzel.) Bullock and in silico identification of its antidiabetic agent. Bioinformatics and Biology Insights, 18. https://doi.org/10.1177/11779322231223857
Babatunde, O. A., & Olusola, A. O. (2024). Gc–Ms Profiling and Biological Activities of Fatty Acid–Rich Plant Fractions. Journal Of Applied Pharmaceutical Science, 14(2), 115–124.
Bamisaye, F. A., Orji, E. E., Ajiboye, B. O., & Oluwajobi, A. O. (2024). Antioxidant activities of aqueous leaf extract of Parquetina nigrescens in high-fat-diet-streptozotocin-induced diabetic male albino rats. Comparative Clinical Pathology, 33(2), 239-246.
Becker, K., Tilley, L., Vennerstrom, J. L., Roberts, D., Rogerson, S., & Ginsburg, H. (2004). Oxidative Stress In Malaria Parasite-Infected Erythrocytes: Host–Parasite Interactions. International Journal For Parasitology, 34(2), 163–189. Https://Doi.Org/10.1016/j.Ijpara.2003.09.011
Burzyńska, A., Wawrzyńczak, A., & Feliczak-Guzik, A. (2025). Systems for Mitochondria-Protective Cosmetic Actives: Opportunities in Post-Oncologic Skin Regeneration. Cosmetics, 13(1), 7.
Cabaço, L. C., Tomás, A., Pojo, M., & Barral, D. C. (2022). The dark side of melanin secretion in cutaneous melanoma aggressiveness. Frontiers in Oncology, 12(May), 1–16. https://doi.org/10.3389/fonc.2022.887366
Corradi, I. D. F., de Souza, E., Sande, D., & Takahashi, J. A. (2018). Correlation between phenolic compounds contents, anti-tyrosinase and antioxidant activities of plant extracts. Chemical Engineering Transactions.
Daramola, O., Oyeyemi, W., Akinola, A., & Raji, Y. (2025). Haematological and biochemical changes in arsenic trioxide-induced toxicity in Wistar rats treated with methanol extract of Parquetina nigrescens leaf. Nigerian Journal of Physiological Sciences : Official Publication of the Physiological Society of Nigeria, 39(1). https://doi.org/10.54548/njps.v37i2.11
Ebrahimi, A., Et Al. (2025). Natural Phenolics as Tyrosinase Inhibitors: Structure–Activity Relationships. Food Chemistry, 430, 137210.
Garbe, C., Forsea, A. M., Amaral, T., Arenberger, P., Autier, P., Berwick, M., Boonen, B., Bylaite, M., del Marmol, V., Dreno, B., Fargnoli, M. C., Geller, A. C., Green, A. C., Greinert, R., Hauschild, A., Harwood, C. A., Hoorens, I., Kandolf, L., Kaufmann, R., ... & Brochez, L. (2024). Skin cancers are the most frequent cancers in fair-skinned populations, but we can prevent them. European Journal of Cancer, 204(114074), 1–5. https://doi.org/10.1016/j.ejca.2024.114074
Gomes, A. R. Q., Cunha, N., Varela, E. L. P., Brígido, H. P. C., Vale, V. V., Dolabela, M. F., ... & Percário, S. (2022). Oxidative stress in malaria: Potential benefits of antioxidant therapy. International Journal of Molecular Sciences, 23(11), 5949. https://doi.org/10.3390/ijms23115949
Hassan, M., Shahzadi, S., & Kloczkowski, A. (2023). Tyrosinase inhibitors naturally present in plants and synthetic modifications of these natural products as anti-melanogenic agents: A review. Molecules, 28(378), 1–13. https://doi.org/10.3390/molecules28010378Hongbao, M. (2017). Resazurin-Based Cytotoxicity Assay Protocol. Methodsx, 4, 128–132.
Hsu, J., Yang, C., & Chen, J. (2022). Antioxidant, anti-α-glucosidase, antityrosinase, and anti-inflammatory activities of bioactive components from Morus alba. Antioxidants, 11. https://doi.org/10.3390/antiox11112222
Huang, L., Tan, L., Lv, Z., Chen, W., & Wu, J. (2024). Pharmacology of bioactive compounds from plant extracts for improving non-alcoholic fatty liver disease through endoplasmic reticulum stress modulation: A comprehensive review. Heliyon, 10. https://doi.org/10.1016/j.heliyon.2024.e25053
Jin, W., Stehbens, S. J., Barnard, R. T., Blaskovich, M. A. T., & Ziora, Z. M. (2024). Dysregulation of tyrosinase activity: A potential link between skin disorders and neurodegeneration. Journal of Pharmacy and Pharmacology, 76(1), 13–22. https://doi.org/10.1093/jpp/rgad107
Kayode, O. T., Ohanaka, N. J., Femi-Olabisi, F. J., Agboola, A. O., & Oyebanji, E. O. (2022). Parquetina nigrescens Species: A Concise Review of Phytochemistry and Pharmacology. Tropical Journal Of Natural Product Research, 6(4), 1–8.
Ko, H. H., & Cho, S. M. (2018). Diterpenoids And Tyrosinase Inhibition. Phytotherapy Research, 32(9), 1764–1772.
Kotepui, M., Mahittikorn, A., Wilairatana, P., Masangkay, F. R., Wangdi, K., & Kotepui, K. U. (2024). Methemoglobin levels in malaria: A systematic review and meta-analysis of its association with Plasmodium falciparum and Plasmodium vivax infections and disease severity. Scientific Reports, 14(1), 3276. https://doi.org/10.1038/s41598-024-53741-6
Kumar U. R., Singh, A., & Biswas, D. (2025). Unraveling the intricate roles of plant secondary metabolites in mitigating bacterial multidrug resistance: Challenges and prospects. Journal of Natural Products. https://doi.org/10.1021/acs.jnatprod.5c00349
Lahlou, M. (2013). The Success of Natural Products In Drug Discovery. Pharmacology & Pharmacy, 4(3), 17–31.
Lajis, N. H., Et Al. (2012). Natural Tyrosinase Inhibitors: Stability and Safety Perspectives. Journal Of Medicinal Plants Research, 6(12), 2345–2352.
Larayetan, R., Ololade, Z. S., Ogunmola, O. O., & Ladokun, A. (2019). Phytochemical constituents, antioxidant, cytotoxicity, antimicrobial, antitrypanosomal, and antimalarial potentials of the crude extracts of Callistemon citrinus. Evidence-Based Complementary and Alternative Medicine, 2019(1), 5410923.
Li, G., & Lou, H. X. (2018). Strategies to Diversify Natural Products for Drug Discovery. Medicinal Research Reviews, 38(4), 1255–1294.
Makler, M. T., Ries, J. M., Williams, J. A., Bancroft, J. E., Piper, R. C., Gibbins, B. L., & Hinrichs, D. J. (1993). Parasite Lactate Dehydrogenase as an Assay for Plasmodium falciparum Drug Sensitivity. American Journal of Tropical Medicine and Hygiene, 48(6), 739–741.
Manap, A. S. A., Lum, Y. K., Ong, L. H., Tang, Y. Q., Gew, L. T., & Chia, A. Y. Y. (2021). Perspective approaches on melanogenesis inhibition. Dermatologica Sinica, 39(1), 1–12. https://doi.org/10.4103/ds.ds_46_20
Moon, K. M., Kwon, E.-B., Lee, B., & Kim, C. Y. (2020). Recent trends in controlling the enzymatic browning of fruit and vegetable products. Molecules, 25(12), 2754. https://doi.org/10.3390/molecules25122754
Ofori, E. A., Garcia-Senosiain, A., Naghizadeh, M., Kana, I. H., Dziegiel, M. H., Adu, B., ... & Theisen, M. (2023). Human blood neutrophils generate ROS through FcγR-signaling to mediate protection against febrile P. falciparum malaria. Communications Biology, 6(1), 743. https://doi.org/10.1038/s42003-023-05091-3
Olaleye, O., Oladipupo, A., Oyawaluja, B., & Coker, H. (2021). Chemical composition, antioxidative and antimicrobial activities of different extracts of the leaves of Parquetina nigrescens (Asclepiadaceae). Polycyclic Aromatic Compounds, 337–349. https://doi.org/10.22034/pcbr.2021.287025.1188
Oyebamiji, O., Awakan, O., Afolabi, B., Oluwadahunsi, S., & Odetola, A. (2025). Phytochemical screening of petroleum ether extract of Xylopia aethiopica. Scientia Africana. https://doi.org/10.4314/sa.v24i3.10
Özgen, R., Sezen Karaoğlan, E., Sevindik, H. G., Hancı, H., & Kazaz, C. (2024). Antimicrobial, antioxidant, and antityrosinase activities of Morina persica L. and its isolated compounds. Molecules, 29(13), 3017.
Panzella, L., & Napolitano, A. (2019). Natural Phenolic Compounds as Tyrosinase Inhibitors. Antioxidants, 8(12), 569.
Patil, R. H., Patil, M. P., & Maheshwari, V. L. (2023). Phytochemistry Of Apocynaceae Members. In Apocynaceae Plants: Ethnobotany, Phytochemistry, Bioactivity and Biotechnological Advances (Pp. 83–104).
Pawłowska, M., Mila-Kierzenkowska, C., Szczegielniak, J., & Woźniak, A. (2023). Oxidative Stress in Parasitic Diseases—Reactive Oxygen Species as Mediators of Interactions between the host and the Parasites. Antioxidants, 13(1), 38. Https://Doi.Org/10.3390/Antiox13010038
Prakash, V. E. D. (2017). Terpenoids as source of anti-inflammatory compounds. Asian Journal of Pharmaceutical and Clinical Research, 10(3), 68–76.
Shaikh, J. R., & Patil, M. K. (2020). Qualitative Tests for Preliminary Phytochemical Screening: an Overview. International Journal of Chemical Studies, 8(2), 603–608.
Shehu, A., Egwemi, O. I., Ajayi, O. A., & Hamid, A. A. (2022). Phytochemical and Antibacterial Evaluations of Chloroform Extract of Mondia whitei (Hook F) Skeels. Tanzania Journal of Science, 48(4), 741-746.
Sui, X., Meng, Z., Dong, T., Fan, X., & Wang, Q. (2023). Enzymatic browning and polyphenol oxidase control strategies. Current Opinion in Biotechnology, 81, 102921. https://doi.org/10.1016/j.copbio.2023.102921Thawabteh, A., Et Al. (2023). Hormonal And Environmental Factors in Hyperpigmentation. Clinical, Cosmetic and Investigational Dermatology, 16, 1021–1034.
Tilkat, E. A., Batibay, H., Yener, I., Yilmaz, P. K., Akdeniz, M., Kaplan, A., ... & Holubec, V. (2021). Determination of enzyme inhibition potential and anticancer effects of Pistacia khinjuk stocks raised in in vitro and in vivo conditions. Agronomy, 11(1), 154
Ty, M. C., Zuniga, M., Götz, A., Kayal, S., Sahu, P. K., Mohanty, A., ... & Rodriguez, A. (2019). Malaria inflammation by xanthine oxidase-produced reactive oxygen species. EMBO Molecular Medicine, 11(8), e9903. https://doi.org/10.15252/emmm.201809903
Vasquez, M., Zuniga, M., & Rodriguez, A. (2021). Oxidative stress and pathogenesis in malaria. Frontiers in Cellular and Infection Microbiology, 11, 768182. https://doi.org/10.3389/fcimb.2021.768182
Wang, X., Kang, W., Li, J., Deng, Z., & Gao, J. (2025). Nisin: harnessing nature’s preservative for the future of food safety and beyond. Critical Reviews in Food Science and Nutrition, 65(34), 9045-9070.
Wawrzyńczak, A. (2023). Uv Radiation and Melanogenesis Pathways. Photodermatology, Photoimmunology & Photomedicine, 39(4), 289–297.
Yadav, K., & Joshi, J. (2024). Biological Activity of Phytochemicals Extracted from Medicinal Plants of Apocynaceae Family. Materials Today: Proceedings.
Yadav, N., & Pandey, A. (2025). Flavonoids as Copper-Chelating Tyrosinase Inhibitors. Journal of Enzyme Inhibition and Medicinal Chemistry, 40(1), 102–110.
Zeitoun, H., Michael‐Jubeli, R., El Khoury, R., Baillet‐Guffroy, A., Tfayli, A., Salameh, D., & Lteif, R. (2020). Skin lightening effect of natural extracts coming from Senegal botanical biodiversity. International Journal of Dermatology, 59(2), 178-183.
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Ekundayo Timi Areh, Yinusa Isah, Abdullahi Shehu

This work is licensed under a Creative Commons Attribution 4.0 International License.




