GASTROPROTECTIVE AND REPARATIVE EFFECTS OF Alstonia boonei AGAINST ASPIRIN-INDUCED GASTROENTEROPATHY IN RAT’S MODEL
DOI:
https://doi.org/10.33003/fjs-2026-1006-4360Keywords:
Alstonia boonei, Aspirin-induced gastroenteropathy, NSAID gastropathyAbstract
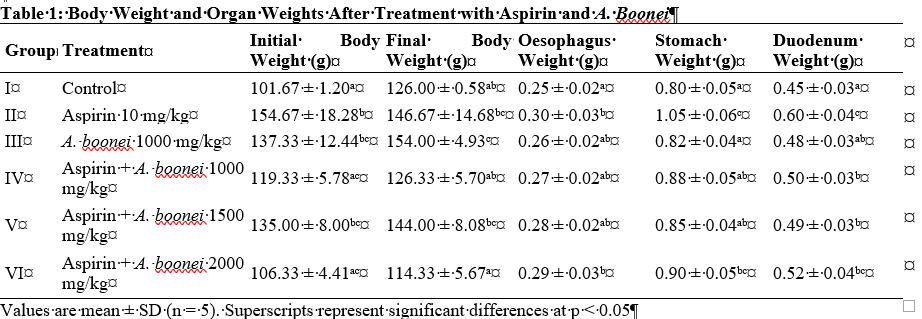
Nonsteroidal anti-inflammatory drugs such as aspirin are widely used but are limited by their potential to induce gastrointestinal injury through oxidative stress, inflammation, and epithelial disruption. This study investigated the protective effects of Alstonia boonei against aspirin-induced gastroenteropathy by evaluating body and organ weights, oxidative stress markers, gross gastric lesions, and histopathological changes in the esophagus, stomach, and duodenum. Rats administered aspirin exhibited reduced weight gain, increased gastric and duodenal organ weights, elevated malondialdehyde levels, depletion of glutathione, and a markedly increased gastric lesion index. These changes were accompanied by severe mucosal alterations, including inflammatory infiltration, glandular distortion, villous disruption, and early erosive damage. Administration of A. boonei alone produced no adverse effects and maintained normal physiological and histological profiles, confirming its safety. Co-treatment with A. boonei resulted in progressive, dose-dependent protection against aspirin-induced injury. Improvements included restoration of weight gain, reduction of organ weight abnormalities, attenuation of oxidative stress, and significant decreases in gross gastric lesions. Histologically, the extract preserved epithelial integrity, reduced inflammatory cell infiltration, and supported normal glandular and villous architecture, with the most pronounced recovery observed at 1500–2000 mg/kg. At these doses, mucosal features approached those of the control group, indicating near-complete structural and biochemical restoration. Overall, the findings demonstrate that A. boonei confers substantial gastroprotective effects through antioxidant and anti-inflammatory mechanisms that stabilize gastrointestinal tissues and counteract aspirin-mediated injury. The extract shows promise as a natural therapeutic agent for mitigating NSAID-induced gastrointestinal damage and warrants further investigation.
References
Adjouzem, C. F., Gilbert, A., Mbiantcha, M., Yousseu Nana, W., Matah Marthe Mba, V., Djuichou Nguemnang, S. F., Tsafack, E. G., & Atsamo, A. D. (2020). Effects of Aqueous and Methanolic Extracts of Stem Bark of Alstonia boonei De Wild. (Apocynaceae) on Dextran Sodium Sulfate-Induced Ulcerative Colitis in Wistar Rats. Evidence-based complementary and alternative medicine: eCAM, 2020, 4918453. https://doi.org/10.1155/2020/4918453
Bhattacharyya, A., Chattopadhyay, R., Mitra, S., & Crowe, S. E. (2014). Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiological reviews, 94(2), 329–354. https://doi.org/10.1152/physrev.00040.2012
Bindu, S., Mazumder, S., & Bandyopadhyay, U. (2020). Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochemical pharmacology, 180, 114147. https://doi.org/10.1016/j.bcp.2020.114147
Bjarnason, I., Scarpignato, C., Holmgren, E., Olszewski, M., Rainsford, K. D., & Lanas, A. (2018). Mechanisms of damage to the gastrointestinal tract from nonsteroidal anti-inflammatory drugs. Gastroenterology, 154(3), 500–514. https://doi.org/10.1053/j.gastro.2017.11.081
Carr, D. F., & Pirmohamed, M. (2018). Biomarkers of adverse drug reactions. Experimental biology and medicine (Maywood, N.J.), 243(3), 291–299. https://doi.org/10.1177/1535370217733425
Chandimali, N., Bak, S. G., Park, E. H., & Lee, S. J. (2025). Free radicals and their impact on health and antioxidant defenses: A review. Cell Death Discovery, 11, 19. https://doi.org/10.1038/s41420-024-02278-8
Chen, Y., Mei, Y. Q., Hou, L., & Li, K. J. (2025). Therapeutic potential of plant-derived natural products against drug-induced liver injury. Frontiers in pharmacology, 16, 1652860. https://doi.org/10.3389/fphar.2025.1652860
Cherrada, N., Chemsa, A. E., Gheraissa, N., Laib, I., Gueboudji, Z., El-Shazly, M., Zaater, A., Abid, A., Sweilam, S. H., Emran, T. B., Nani, S., Benamor, B., Ghemam Amara, D., Atoki, A. V., & Messaoudi, M. (2024). Gastroprotective Efficacy of North African Medicinal Plants: A Review on Their Therapeutic Potential for Peptic Ulcers. Food science & nutrition, 12(11), 8793–8824. https://doi.org/10.1002/fsn3.4536
Cherrada, N., Chemsa, A. E., Gheraissa, N., Laib, I., Gueboudji, Z., El-Shazly, M., Zaater, A., Abid, A., Sweilam, S. H., Emran, T. B., Nani, S., Benamor, B., Ghemam Amara, D., Atoki, A. V., & Messaoudi, M. (2024). Gastroprotective Efficacy of North African Medicinal Plants: A Review on Their Therapeutic Potential for Peptic Ulcers. Food science & nutrition, 12(11), 8793–8824. https://doi.org/10.1002/fsn3.4536
Hijos-Mallada, G., Sostres, C., & Gomollón, F. (2022). NSAIDs, gastrointestinal toxicity and inflammatory bowel disease. Gastroenterología y Hepatología (English Edition), 45(3), 215-222. https://doi.org/10.1016/j.gastre.2021.06.002
Hladkykh, F., & Chyzh, M. (2021). Nonsteroidal anti-inflammatory drugs: A modern understanding of the mechanisms of damage to the digestive tract, the shortcomings of pathogenetic drugs and prospects for biological therapy of NSAID-induced esophagogastroenterocolonopathy. Gastroenterology, 54(4), 253–266. https://doi.org/10.22141/2308-2097.54.4.2020.216714
Iwamoto, J., Saito, Y., Honda, A., & Matsuzaki, Y. (2013). Clinical features of gastroduodenal injury associated with long-term low-dose aspirin therapy. World journal of gastroenterology, 19(11), 1673–1682. https://doi.org/10.3748/wjg.v19.i11.1673
Kakizaki, F., Miyazaki, T., Ueda, H., Iwamoto, J., Honda, A., & Ikegami, T. (2025). Small intestinal bacteria accelerate aspirin-induced small intestinal injuries. Experimental and Molecular Pathology, 143, 104984. https://doi.org/10.1016/j.yexmp.2025.104984
Lavie, C., Howden, C. W., Scheiman, J., & Tursi, J. (2017). Upper gastrointestinal toxicity associated with long-term aspirin therapy: Consequences and prevention. Current Problems in Cardiology, 42(5), 146–166. https://doi.org/10.1016/j.cpcardiol.2017.01.006
Martins-Gomes, C., Nunes, F. M., & Silva, A. M. (2024). Natural products as dietary agents for the prevention and mitigation of oxidative damage and inflammation in the intestinal barrier. Antioxidants, 13(1), 65. https://doi.org/10.3390/antiox13010065
Matsui, H., Shimokawa, O., Kaneko, T., Nagano, Y., Rai, K., & Hyodo, I. (2011). The pathophysiology of non-steroidal anti-inflammatory drug (NSAID)-induced mucosal injuries in stomach and small intestine. Journal of clinical biochemistry and nutrition, 48(2), 107–111. https://doi.org/10.3164/jcbn.10-79
Mollica, A., Zengin, G., Sinan, K. I., Marletta, M., Pieretti, S., Stefanucci, A., Etienne, O. K., Jekő, J., Cziáky, Z., Bahadori, M. B., Picot-Allain, C., & Mahomoodally, M. F. (2022). A Study on Chemical Characterization and Biological Abilities of Alstonia boonei Extracts Obtained by Different Techniques. Antioxidants (Basel, Switzerland), 11(11), 2171. https://doi.org/10.3390/antiox11112171
Mugale, M. N., Dev, K., More, B. S., Mishra, V. S., Washimkar, K. R., Singh, K., Maurya, R., Rath, S. K., Chattopadhyay, D., & Chattopadhyay, N. (2024). A comprehensive review on preclinical safety and toxicity of medicinal plants. Clinical Complementary Medicine and Pharmacology, 4(1), 100129. https://doi.org/10.1016/j.ccmp.2024.100129
Muro, P., Zhang, L., Li, S., Zhao, Z., Jin, T., Mao, F., & Mao, Z. (2024). The emerging role of oxidative stress in inflammatory bowel disease. Frontiers in endocrinology, 15, 1390351. https://doi.org/10.3389/fendo.2024.1390351
Okorie, N. H., Mbah, C. J., Ugodi, G. W., & Magbo, C. C. (2022). Investigation into the gastro-protective properties of Alstonia boonei (de wild) leaves and stem bark. Tropical Journal of Natural Product Research, 6(3), 403–407. https://tjnpr.org/viewarticle.aspx?articleid=2258
Olanlokun, J. O., Olowofolahan, A. O., Bodede, O., Adegbuyi, A. T., Prinsloo, G., Steenkamp, P., & Olorunsogo, O. O. (2021). Anti-Inflammatory Potentials of the n-Hexane Fraction of Alstonia boonei Stem Bark in Lipopolysaccharide-Induced Inflammation in Wistar Rats. Journal of inflammation research, 14, 3905–3920. https://doi.org/10.2147/JIR.S304076
Organisation for Economic Co-operation and Development (OECD). (2022, June 30). Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure. OECD Guidelines for the Testing of Chemicals, Section 4. https://www.oecd.org/en/publications/2022/06/test-no-425-acute-oral-toxicity-up-and-down-procedure_g1gh2953.html
Oyebode, O. A., Erukainure, O. L., Ibeji, C. U., Koorbanally, N. A., & Islam, M. S. (2019). Phytochemical constituents, antioxidant and antidiabetic activities of different extracts of the leaves, stem and root barks of Alstonia boonei: An in vitro and in silico study. Ethnobotany & Pharmacology, 166(4), 444–456. https://doi.org/10.1080/23818107.2019.1624980
Park, J. U., Kang, J. H., Rahman, M. A. A., Hussain, A., Cho, J. S., & Lee, Y. I. (2019). Gastroprotective Effects of Plants Extracts on Gastric Mucosal Injury in Experimental Sprague-Dawley Rats. BioMed research international, 2019, 8759708. https://doi.org/10.1155/2019/8759708
Sohail, R., Mathew, M., Patel, K. K., Reddy, S. A., Haider, Z., Naria, M., Habib, A., Abdin, Z. U., Razzaq Chaudhry, W., & Akbar, A. (2023). Effects of Non-steroidal Anti-inflammatory Drugs (NSAIDs) and Gastroprotective NSAIDs on the Gastrointestinal Tract: A Narrative Review. Cureus, 15(4), e37080. https://doi.org/10.7759/cureus.37080
Takeuchi K. (2012). Pathogenesis of NSAID-induced gastric damage: importance of cyclooxygenase inhibition and gastric hypermotility. World journal of gastroenterology, 18(18), 2147-2160. https://doi.org/10.3748/wjg.v18.i18.2147
Tawfik, A. G., Gomez-Lumbreras, A., Del Fiol, G., Kawamoto, K., Trinkley, K. E., Reese, T., Jones, A., & Malone, D. C. (2025). Nonsteroidal anti-inflammatory drugs and risk of gastrointestinal bleeding: A systematic review and meta-analysis. Clinical Pharmacology & Therapeutics. Advance online publication. https://doi.org/10.1002/cpt.70054
Tulassay, Z., & Herszényi, L. (2010). Gastric mucosal defense and cytoprotection. Best practice & research. Clinical gastroenterology, 24(2), 99–108. https://doi.org/10.1016/j.bpg.2010.02.006
Uroko, R., Sangodare, R., Onyeabo, C., Agbafor, A., Uchenna, O., Nwuke, C., & Asadu, C. (2020). Investigation of antioxidant composition and antioxidative activities of ethanol extract of Alstonia boonei. Nigerian Journal of Pharmaceutical Research, 16(1), 71-80. https://doi.org/10.4314/njpr.v16i1.8
Wallace J. L. (2013). Mechanisms, prevention and clinical implications of nonsteroidal anti-inflammatory drug-enteropathy. World journal of gastroenterology, 19(12), 1861-1876. https://doi.org/10.3748/wjg.v19.i12.1861
Wallace, J. L. (2012). NSAID gastropathy and enteropathy: Distinct pathogenesis likely necessitates distinct prevention strategies. British Journal of Pharmacology, 165(1), 67–74. https://doi.org/10.1111/j.1476-5381.2011.01509.x
Zhou, J., Li, N., Li, X., Ye, J., Wang, M., & Sun, G. (2025). Review on recent advancements in understanding acetylsalicylic acid-induced gastrointestinal injury: Mechanisms, medication, and dosage refinement. Naunyn-Schmiedeberg's Archives of Pharmacology, 398(11), 3297–3320. https://doi.org/10.1007/s00210-024-03521-w
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Blessing Emosho Ogeyemhe, Efosa Bolaji Odigie

This work is licensed under a Creative Commons Attribution 4.0 International License.




