ASSESSMENT OF ACUTE AND SUB-ACUTE TOXICITY STUDY OF GARCINA KOLA HECKEL SEEDS IN WISTAR RAT
DOI:
https://doi.org/10.33003/fjs-2026-1002-4660Keywords:
Garcina kola Heckel seed, weight, Haematology, Serum chemistryAbstract
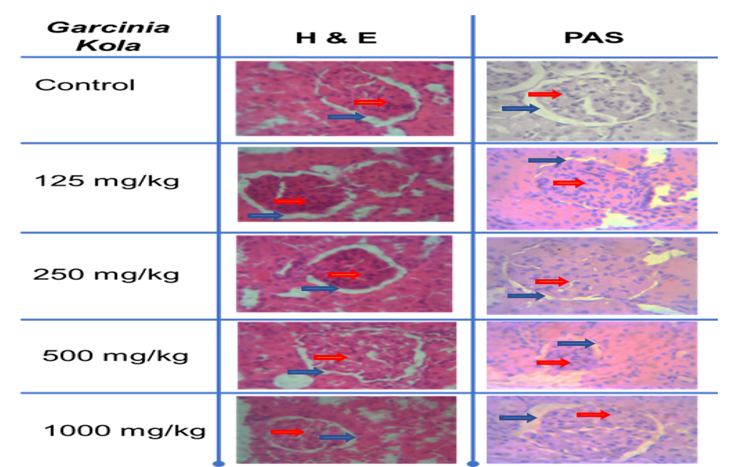
Garcina kola Heckel have been used in the treatment of many diseases such as diabetes mellitus, erectile dysfunction, chronic cough and liver disease. This study aimed at investigating acute and sub-acute toxic effects of Garcina kola Heckel seed on physiological, biochemical and histopathological parameters of Wistar rat. Acutely, nine rats (n=3) were used as; A (control); B (1000 mg/kg) and C (2000 mg/kg). Sub-acutely, twenty-five rats grouped; A-E (n=5). A (control), B (125 mg/kg); C (250 mg/kg); D (500 mg/kg) and E (1000 mg/kg). Rats were treated with the extract consecutively for 21 days. Garcina kola Heckel seed is safe acutely at tested dose (1000 and 2000 mg/kg) as no mortality/visible toxic reaction was observed. The extract showed non-significant weight gain (day 7) but reduced significantly across the 21 days with most significant reduction in 1000 mg/kg. Haematological result showed that PVC, RBC, haemoglobin and platelets had no significant (P>0.05) effect unlike white blood cell and its differentia (neutrophils, lymphocytes and monocytes) which decrease significantly (P <0.05). Creatinine increased significantly (P<0.05) all treated groups. Albumin, ALT, ALP decrease significantly. AST statistically not significant except group 2 while Total Bilirubin and Total protein decreased significantly compared to the control. Conclusively, Garcina kola Heckel seed is safe acutely at administered dosages. Weight was significantly reduced in the course of the treatment while altering the haematological and biochemical parameters. Histopathological result showed that no significant changes were observed using both assessments ensuring a further indication of the safety of the G. kola extract.
References
Abarikwu, S. O., Otuechere, C. A., Ekor, M., Monwuba, K., & Osobu, D. (2017). Rutin ameliorates Garcinia kola–induced hepatotoxicity in Wistar rats by suppressing oxidative stress and inflammatory responses. Toxicology Reports, 4, 79–89.
Ahad, B., Shahri, W., Rasool, H., Reshi, Z. A., Rasool, S., & Hussain, T. (2021). Medicinal plants and herbal drugs: An overview. In Medicinal and Aromatic Plants: Healthcare and Industrial Applications (pp. 1–40).
Aremu, A., Oridupa, O. A., Akorede, G. J., Olatumji, A. O., Basiru, A., Ahmed, A. O., & Raufu, I. A. (2022). Safety evaluation of Lawsonia inermis on physiological, andrological and haematological parameters of male Wistar rats.
Chintada, V., & Golla, N. (2025). Exploring the therapeutic potential of bioactive compounds from plant sources. In Biotechnological Intervention in Production of Bioactive Compounds: Biosynthesis, Characterization and Applications (pp. 229–247). Springer Nature Switzerland.
Dogara, A. M., Hamad, S. W., Hama, H. A., Bradosty, S. W., Kayfi, S., Al-Rawi, S. S., & Lema, A. A. (2022). Biological evaluation of Garcinia kola Heckel. Advances in Pharmacological and Pharmaceutical Sciences, 2022, 3837965.
Emmanuel, O., Uche, M. E., Dike, E. D., Etumnu, L. R., Ugbogu, O. C., & Ugbogu, E. A. (2022). A review on Garcinia kola Heckel: Traditional uses, phytochemistry, pharmacological activities, and toxicology. Biomarkers, 27(2), 101–117.
Enaiho, S. O., & Ekhoye, E. I. (2025). Modulation of dexamethasone-induced testicular dysfunction by Garcinia kola seed-derived kolaviron in rats. FUOYE Journal of Biomedical Research, 2(1).
Galam, N. Z., Gambo, I. M., Habeeb, A. A., & Shugaba, A. I. (2013). Effect of aqueous extract of Garcinia kola seed on liver histology. Journal of Natural Sciences Research, 3(1), 81–87.
Hudson, A., Lopez, E., Almalki, A. J., Roe, A. L., & Calderón, A. I. (2018). A review of the toxicity of compounds found in herbal dietary supplements. Planta Medica, 84(9–10), 613–626.
Jităreanu, A., Trifan, A., Vieriu, M., Caba, I. C., Mârțu, I., & Agoroaei, L. (2022). Current trends in toxicity assessment of herbal medicines: A narrative review. Processes, 11(1), 83.
Malann, Y. D. D. (2018). Effects of Mangifera indica extract on haematological and histopathological indices in laboratory mice.
Nwachukwu, K. C., Ibe, C., Nwachukwu, N. C., & Ugbogu, O. C. (2022). Haematological, biochemical and histopathological assessment of the toxicity potential of seed kernel extracts of Mangifera indica Linn. varieties in mice. Discovery, 58(322), 1117–1127.
Nwozo, O. S., Effiong, E. M., Aja, P. M., & Awuchi, C. G. (2023). Antioxidant, phytochemical and therapeutic properties of medicinal plants: A review. International Journal of Food Properties, 26(1), 359–388.
Obeta, U. M., Alexander, L., Ejinaka, O. R., Ekpe, C. A., Okey-Ndeche, U., & Joy-Amobi, C. (2023). Comparative assessment of medicinal importance of bitter kola and walnut. Skin, 1, 8.
Ojatula, A. O., & Ezenwa, K. C. (2023). Sub-acute toxicity effects of Garcinia kola on serum electrolytes, haematological indices, visceral organ weights and histopathological profiles in Wistar rats. Science World Journal, 18(2), 283–289.
Porras, G., Chassagne, F., Lyles, J. T., Marquez, L., Dettweiler, M., & Salam, A. M. (2021). Ethnobotany and the role of plant natural products in antibiotic drug discovery. Chemical Reviews, 121(6), 3495–3560.
Reyes, C. M., & Cornelis, M. C. (2018). Caffeine in the diet: Country-level consumption and guidelines. Nutrients, 10(11), 1772.
Saggar, S., Mir, P. A., Kumar, N., Chawla, A., Uppal, J., & Kaur, A. (2022). Traditional and herbal medicines: Opportunities and challenges. Pharmacognosy Research, 14(2), 107–114.
Sahil, Sharma, M., Das, A., Saxena, S., & Rather, S. A. (2021). Toxicity: Its assessment and remediation in important medicinal plants. In Medicinal and Aromatic Plants: Healthcare and Industrial Applications (pp. 571–591). Springer International Publishing.
Schultz, F., & Garbe, L. A. (2023). How to approach a study in ethnopharmacology? Pharmacology Research & Perspectives, 11(4), e01109.
Sroor, F. M., Basyouni, W. M., Aly, H. F., Younis, E. A., Mahrous, K. F., & Haroun, A. A. (2022). Biochemical and histopathological studies of sulfonylurea derivative as a new chemotherapeutic agent against liver cancer. Applied Biological Chemistry, 65(1), 68.
Tamber, S. S., Bansal, P., Sharma, S., Singh, R. B., & Sharma, R. (2023). Biomarkers of liver diseases. Molecular Biology Reports, 50(9), 7815–7823.
Tauchen, J., Frankova, A., Manourova, A., Valterova, I., Lojka, B., & Leuner, O. (2023). Garcinia kola: A critical review on chemistry and pharmacology of an important West African medicinal plant. Phytochemistry Reviews, 22(5), 1305–1351.
Tordzagla, N., Blay, E. E., & Mohammed, B. S. (2025). Antioxidant and antidiabetic properties and safety profile of methanolic extract of Garcinia kola seed. Investigational Medicinal Chemistry and Pharmacology, 8(1), 2–2.
Yogom, B. T., Avana-Tientcheu, M. L., Mboujda, M. F. M., Momo, S. T., Fonkou, T., Tsobeng, A., … Duminil, J. (2020). Ethnicity differences in uses and management practices of bitter kola (Garcinia kola) in Cameroon. Economic Botany, 74(4), 429–444.
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Adeshina Salmat Aremu, Otuofu Amali Muhammed, Abdulmalik Aliyu, Abdulfatai Aremu

This work is licensed under a Creative Commons Attribution 4.0 International License.




