PROTECTIVE EFFECTS OF RUTIN AGAINST SODIUM ARSENITE-INDUCED HEMATOLOGICAL ALTERATIONS AND OXIDATIVE STRESS IN WISTAR RATS
DOI:
https://doi.org/10.33003/fjs-2026-1002-4569Keywords:
Sodium arsenite, Rutin, Oxidative stress, Hematological alterations, Wistar rats, Neutrophil-to-lymphocyte ratio, Antioxidant enzymes, Lipid peroxidationAbstract
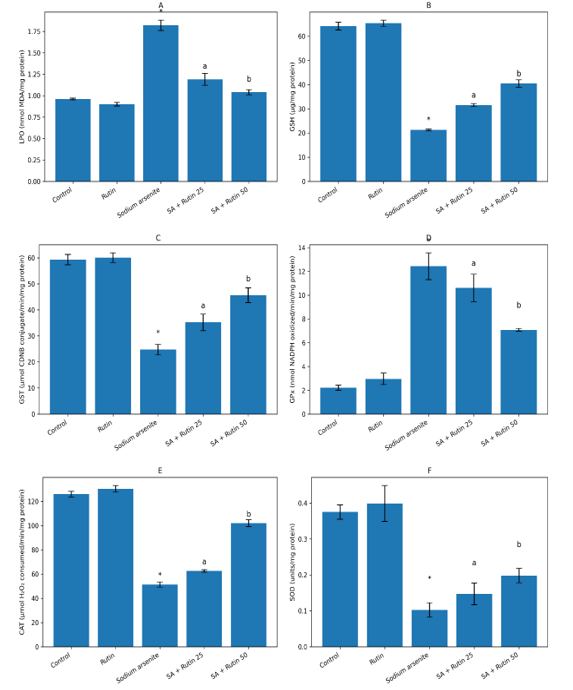
Sodium arsenite (NaAsO₂) is a pervasive environmental toxicant known to induce severe hematological alterations and systemic oxidative stress. Rutin, a natural bioflavonoid, exhibits potent antioxidant and anti-inflammatory properties, suggesting potential protective effects against such toxicity. This study investigated the ameliorative effects of Rutin on sodium arsenite-induced hematological changes and oxidative stress in Wistar rats. Thirty-five rats were divided into five groups (n=7): Group A (Control), Group B (Rutin alone, 50 mg/kg), Group C (Sodium Arsenite alone), Group D (Sodium Arsenite + Rutin 25 mg/kg), and Group E (Sodium Arsenite + Rutin 50 mg/kg). After the experimental period, hematological indices (RBC, Hb, WBC, differential counts, Neutrophil-Lymphocyte Ratio) and oxidative stress markers (Lipid Peroxidation/LPO, Glutathione/GSH, Glutathione S-transferase/GST, Glutathione Peroxidase/GPx, Catalase/CAT, Superoxide Dismutase/SOD) were analyzed. Sodium arsenite exposure (Group C) caused significant (p<0.05) hematological toxicity, evidenced by decreased RBC count and Hb, alongside increased WBC count, neutrophilia, lymphocytopenia, and elevated NLR. Concurrently, it induced oxidative stress by significantly increasing LPO and depleting GSH, GST, GPx, CAT, and SOD levels. Co-treatment with Rutin, particularly at 50 mg/kg (Group E), demonstrated a significant dose-dependent reversal of these effects. Rutin treatment notably restored RBC and Hb levels, normalized WBC count and differentials, and improved the NLR. Furthermore, it significantly (a,b p<0.05) attenuated oxidative damage by reducing LPO and enhancing the levels of all measured antioxidant markers compared to the arsenic-only group. The findings demonstrate that Rutin possesses a potent protective effect against sodium arsenite-induced hematological dysregulation and oxidative stress in Wistar rats.
References
Aebi, H. (1984). Catalase in vitro. Methods in Enzymology, 105, 121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Al-Dhabi, N. A., Arasu, M. V., Park, C. H., & Park, S. U. (2020). An up-to-date review of rutin and its biological and pharmacological activities. EXCLI Journal, 19, 1569–1586. https://doi.org/10.17179/excli2020-2679
Beutler, E., Duron, O., & Kelly, B. M. (1963). Improved method for the determination of blood glutathione. Journal of Laboratory and Clinical Medicine, 61, 882–888.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Analytical Biochemistry, 72(1–2), 248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Enogieru, A. B., Haylett, W., Hiss, D. C., Bardien, S., & Ekpo, O. E. (2018). Rutin as a potent antioxidant: Implications for neurodegenerative disorders. Oxidative Medicine and Cellular Longevity, 2018, 6241017. https://doi.org/10.1155/2018/6241017
Flora, S. J. S. (2011). Arsenic-induced oxidative stress and its reversibility. Free Radical Biology and Medicine, 51(2), 257–281. https://doi.org/10.1016/j.freeradbiomed.2011.04.008
Ganeshpurkar, A., & Saluja, A. K. (2017). The pharmacological potential of rutin. Saudi Pharmaceutical Journal, 25(2), 149–164. https://doi.org/10.1016/j.jsps.2016.04.025
Habig, W. H., Pabst, M. J., & Jakoby, W. B. (1974). Glutathione S-transferases: The first enzymatic step in mercapturic acid formation. Journal of Biological Chemistry, 249(22), 7130–7139.
Hughes, M. F., Beck, B. D., Chen, Y., Lewis, A. S., & Thomas, D. J. (2011). Arsenic exposure and toxicology: A historical perspective. Toxicological Sciences, 123(2), 305–332. https://doi.org/10.1093/toxsci/kfr184
Jomova, K., Jenisova, Z., Feszterova, M., Baros, S., Liska, J., Hudecova, D., Rhodes, C. J., & Valko, M. (2011). Arsenic: Toxicity, oxidative stress and human disease. Journal of Applied Toxicology, 31(2), 95–107. https://doi.org/10.1002/jat.1649
Marklund, S., & Marklund, G. (1974). Involvement of the superoxide anion radical in the auto-oxidation of pyrogallol and a convenient assay for superoxide dismutase. European Journal of Biochemistry, 47(3), 469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Mazumder, D. N. G. (2005). Effect of chronic intake of arsenic-contaminated water on liver. Toxicology and Applied Pharmacology, 206(2), 169–175. https://doi.org/10.1016/j.taap.2004.11.017
Ohkawa, H., Ohishi, N., & Yagi, K. (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry, 95(2), 351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Paglia, D. E., & Valentine, W. N. (1967). Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. Journal of Laboratory and Clinical Medicine, 70, 158–169.
Ratnaike, R. N. (2003). Acute and chronic arsenic toxicity. Postgraduate Medical Journal, 79(933), 391–396. https://doi.org/10.1136/pmj.79.933.391
Zahorec, R. (2021). Neutrophil-to-lymphocyte ratio: Past, present and future perspectives. Bratislavské Lekárske Listy, 122(7), 474–488. https://doi.org/10.4149/BLL_2021_078
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Omorede Ikponmwosa-Eweka, Ikenna Chukwuemeka Maduako, Aisosa Bliss Ohenhen, Raphael Aima Amiebenomo

This work is licensed under a Creative Commons Attribution 4.0 International License.




