ANTIFUNGAL EFFECTS OF Azadirachta indica AND OCIMUM GRATISSIMUM ON DERMATOPHYTES
DOI:
https://doi.org/10.33003/fjs-2026-1005-4876Keywords:
Azadirachta indica, Minimum inhibitory concentration (MIC), Ocimum gratissimum, Percentage reduction in radial growth (%RRG)Abstract
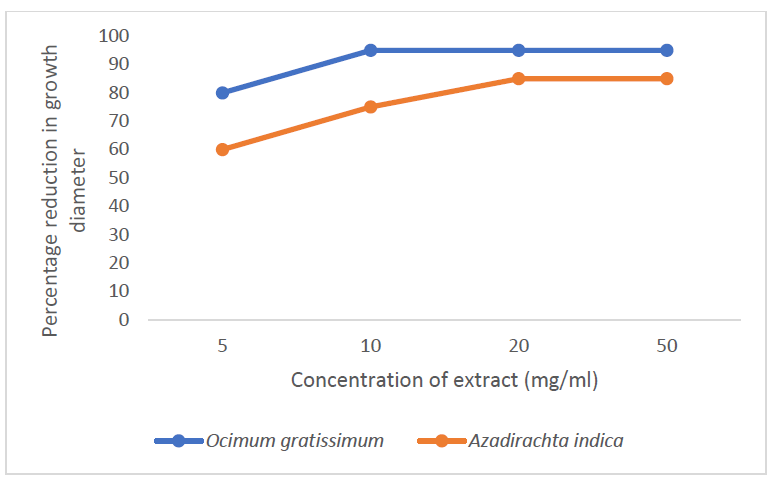
Dermatophytes pose a global burden with a worldwide prevalence of 20 - 25%. This study was aimed at the evaluation of antifungal effects of leaf extracts of Azadirachta indica and Ocimum gratissimum on dermatophytes. Forty nine skin scrapings were obtained from patients at the Skin Disease Clinic, Lagos University Teaching Hospital Annex, Yaba. Dermatophytes were isolated on Sabouraud dextrose agar and identified by microscopic and biochemical methods. The aqueous and ethanolic extractions of the plant leaves was done by maceration and Soxhlet method respectively. The antifungal activity was evaluated using the broth dilution method. Trichophyton mentagrophytes had the highest percentage occurrence of 43.24, followed by T. equinum (27.03), and the lowest was 5.41 for Microsporum gypseum. The aqueous extracts of both plants completely inhibited all the dermatophytes at 250mgml. The minimum inhibitory concentration (MIC) of A. indica was 125mg/ml against all the dermatophytes. However, for O. gratissimum, the MIC was 63mg/ml against T. equinum, T. verrucosum and 125 mg/ml against T. mentagrophytes. For ethanol extract of A. indica, the highest percentage reduction in radial growth (%RRG) was 94.84 at 50 mg/ml against T. equinum and the lowest %RRG was 0.33 at 5 mg/ml against E. fIoccosum. For ethanol extract of O. gratissimum, the highest %RRG was 95.71 at 50 mg/ml against T. verrucosum and the lowest was 23.6 at 5 mg/ml against M. gypseum even as E. fIoccosum was not inhibited at that concentration. The results indicated the effectiveness of studied plant extracts in controlling the growth of dermatophytes.
References
Abubakar, N., & Sandamu, H. S. (2026). Antibacterial activity of Allium sativum (galic) against Neisseria gonorrhoeae and Treponema pallidum. FUDMA Journal of Sciences, 10(1): 253 – 258.
Adejumo T.O., & Bamidele, B. S. (2009). Control of dermatophyte-causing agents (Trichophyton mentagrophytes and Trichophyton rubrum) using six medicinal plants. Journal of Medicinal Plants Research, 3(11): 906 - 913.
Aguguo, K., Umedum, C. U., Nzeagwu, M. O., Opara, N. K., Onyema, C. T., Okorie, C. C., Umejiego, I. B., Korie, M. C., Onwubuche, B. C., & Uzoho, I. J. (2020). Antifungal activity of two plant extracts against fungi isolated from poultry droppings in Owerri, Imo State, Nigeria. Nigerian Journal of Microbiology, 34(1): 5070 – 5079.
Akinnibosun, H. A., Rotimi J., & Aigbogun, O. C. (2009). "Larvicidal activity of leaf extracts of neem- Azadirachta indica A. Juss. (Family: Meliaceae) against mosquito larvae of Aedes aegypti Linn. (Diptera Culicidae). Plant Archives, 9(1): 509 - 513.
Ansari, S., Ahmadi, B., Norouzi, M., Ansari, Z., Afsarian, M. H., Lotfali, E., & Rezaei-Matehkolaei, A. (2019). Epidermophyton fIoccosum: nucleotide sequence analysis and antifungal susceptibility testing of 40 clinical isolates. Journal of Medical Microbiology, 68(11): 1655 - 1663.
Babayi, H., Kolo, I., Okogun, J., & Ijah, U. J. J. (2004). The antimicrobial activities of methanolic extracts of Eucalyptus camaldulensis and Terminalia catappa against some pathogenic microorganisms. Biokemistri, 16(2): 106 – 111.
Bhosale, N. K., Prabha, R., Munuswamy, R., Pramodhini, S., & Easow, J. M. (2022). A comparative study on the phenotypic versus molecular identification of clinical dermatophytes. Journal of Pure and Applied Microbiology, 16(2): 1138 - 1146.
Chandler, D. (2022). Direct microscopy in the dermatology clinic: enhancing the management of skin infections and infestations. Clinical Experimental Dermatology, 47(6): 1023 – 1029.
Egwuatu, T. O., Ukhureigbe, O. M., Ojo, S. K. S., Ogeneh, B. O., Osanyinlusi, S. A., & Ajayi, O. E. (2019). Risk factors associated with Tinea capitis among pupils in Lagos, Ekiti and Ogun States, Nigeria. Nigerian Journal of Microbiology, 33(1): 4380 – 4386.
Geddes‐McAlister, J., & Shapiro, R. S. (2019). New pathogens, new tricks: emerging, drug‐resistant fungal pathogens and future prospects for antifungal therapeutics. Annals of the New York Academy of Sciences, 1435(1):.57 - 78.
Iwuagwu, C. C., Onejeme, F. C., Ononuju, C. C., Umechuruba, C. I., & Nwogbaga, A. C. (2018). Effects of plant extracts and synthetic fungicides on the radial growth of Phoma oryzae on rice (Oryza sativa L) in some rice growing areas of South Eastern Nigeria. Journal of Plant Pathology and Microbiology, 9(12): 468 - 473.
Izah, S. C. (2018). Some determinant factors of antimicrobial susceptibility pattern of plant extracts. Research and Review Insights, 2(3): 1 – 4.
Kator, L., & Hosea, Z. Y. (2018). In vitro potential of aqueous extracts of plant leaves to inhibit pathogenic fungi. Asian Journal of Research in Biochemistry, 2(3):1 - 18.
Kaur, S., & Gill, P. K. (2023). In-vitro anti-dermatophytic activity of different medicinal plant extracts. International Journal of Indigenous Herbs and Drugs, 8(5): 27 - 32.
Koba, K., Poutouli, P. W., Raynaud, C., & Sanda, K. (2009). Antifungal activity of the essential oils from Ocimum gratissimum L. grown in Togo. Journal of Scientific Research, 1(1): 164 – 171.
Lavanya, V. (2013). Isolation and identification of fungi causing dermatophytosis in a tertiary care hospital in bagalkot: Doctoral dissertation, Rajiv Gandhi University of Health Sciences (India). ProQuest Dissertations Publishing, 30559268.
Mahesh, B., & Satish S. (2008). Antimicrobial activity of some important medicinal plants against plant and human pathogens. World Journal of Agricultural Sciences, 4 (5): 839 – 843.
.Mbakwem-Aniebo, C., Onianwa, O., & Okonko, I. O. (2012). Effects of Ocimum gratissimum leaves on common dermatophytes and causative agent of Pityriasis Versicolor in Rivers State, Nigeria. Journal of Microbiology Research, 2(4): 108 – 113.
Mbata, T. I., Duru, M. C., & Onwumelu, H. A. (2009). Antibacterial activity of crude seed extracts of Buchholzia coriacea E. on some pathogenic bacteria. Journal of Developmental Biology and Tissue Engineering, 1(1): 001 - 005.
Miethke, M., Pieroni, M., Weber, T., Brönstrup, M., Hammann, P., Halby, L., Arimondo, P. B., Glaser, P., Aigle, B., Bode, H. B., & Moreira, R. (2021). Towards the sustainable discovery and development of new antibiotics. Nature Reviews Chemistry, 5(10): 726 - 749.
Moskaluk, A. E., & VandeWoude, S. (2022). Current topics in dermatophyte classification and clinical diagnosis. Pathogens, 11(9): 957 – 963.
Nweze, E. I., & Eke, I. E. (2018). Dermatophytes and dermatophytosis in the eastern and southern parts of Africa. Medical Mycology, 56(1): 13 - 28.
Okigbo, R. N., Okeke, J. J., & Madu, N. C. (2010). "Larvicidal effects of Azadirachta indica, Ocimum gratissimum and Hyptis suaveolens against mosquito larvae. Journal of Agricultural Technology, 6(4): 703-719: 703-719.
Penecilla, G. L., & Magno, C. P. (2011). Antibacterial activity of extracts of twelve common medicinal plants in Philipines. Journal of Medicinal Plants Research, 5 (16): 3975 - 3981.
Priyanka, C., Shivika, S., & Vikas, S. (2018). Ocimum gratissimum: A review on ethnomedicinal properties, phytochemical constituents, and pharmacological profile. In: Kumar, N. (ed) Biotechnological approaches for medicinal and aromatic plants, (pp.251-270). Springer Nature.
Rodrigues, M. P., Astoreca, A. L., Oliveira, Á. A. D., Salvato, L. A., Biscoto, G. L., Keller, L. A. M., Rosa, C. A. D. R., Cavaglieri, L. R., Azevedo, M. I. D., and Keller, K. M. (2019). In vitro activity of neem (Azadirachta indica) oil on growth and ochratoxin A production by Aspergillus carbonarius isolates. Toxins, 11(10):.579 - 591.
Rubinchik, E., & Pasetka, C. (2010). Ex vivo skin infection model. In: Giuliani, A., Rinaldi, A. (eds) Antimicrobial Peptides: Methods in Molecular Biology, vol. 618 (pp.359-369). Humana Press, Totowa, NJ..
Sánchez-Bautista, A., Coy, J., García-Shimizu, P., & Rodríguez, J. C. (2018). From CLSI to EUCAST guidelines in the interpretation of antimicrobial susceptibility: What is the effect in our setting? Enfermedades infecciosasy microbiologia clinica (English ed.), 36(4): 229 - 232.
Segal E., & Elad, D. (2021). Human and zoonotic dermatophytoses: epidemiological aspects. Frontiers in Microbiology, 6(12): 713 - 532.
Varaprasad, B., Rajesh, K. C., Varahalarao, V., & Prasanth, K. K. (2009). Medicinal plant as alternative biocontrol agents in the control of seed borne pathogen Macrophomina phaseolina. Varaprasad et al., 2009 of Pharmacy Research, 2(6): 1045 – 1048.
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Adebayo Joseph Grillo, Oluchi Ukhureigbe, Louis Osayenum Egwari, Nurudeen Ayoade Olasupo, Bolanle Adeniyi

This work is licensed under a Creative Commons Attribution 4.0 International License.




