ANTIBIOGRAM PROFILE AND VIRULENCE GENETIC DETERMINANTS OF NON-TYPHOIDAL Salmonella SPECIES ISOLATED FROM THE FAECES OF CHICKENS IN OTA, OGUN STATE, NIGERIA
DOI:
https://doi.org/10.33003/fjs-2026-1005-4832Keywords:
Antimicrobial resistance, Virulence, Non-typhoidal Salmonella, Poultry, NigeriaAbstract
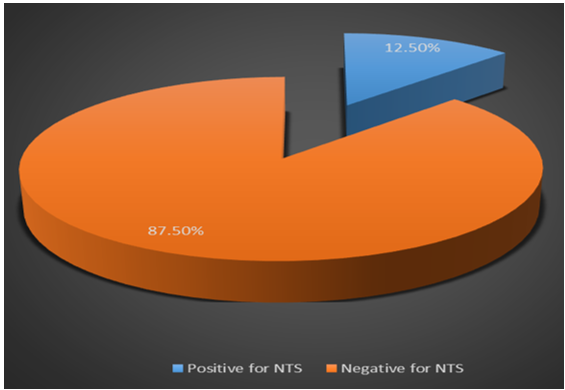
Some healthy chickens harbour Salmonella serovars, which could potentially contaminate raw animal products during processing, and eventually transferred to humans. This study was aimed, at determining the antibiotic resistance pattern and genetic virulence determinants that encode invasion A protein (invA), flagella biosynthetic protein (fliR) and tetrathionate reductase (ttrA) in Salmonella enterica serovars, isolated from the faeces of chickens. Two hundred faecal samples were obtained from a commercial layer poultry farm, and 25 presumptive non-typhoidal strains were recovered. Antibiotic susceptibility testing (AST) was conducted, by the disc diffusion method, and virulence genes were detected by polymerase chain reaction (PCR). The highest resistance among the non-typhoidal Salmonella in this study was to ampicillin (32 %); however, a high proportion of the isolates were susceptible to chloramphenicol (96 %). Out of the 25 serovars, 5 (20 %) exhibited multidrug resistance to 3 or more classes of antibiotics. The invA gene was confirmed in all the 25 (100 %) Salmonella strains, while fliR and ttrA genes were detected in 9 (36 %) and 7 (28 %) of the isolates, respectively. The Salmonella serovars and multidrug-resistant (MDR) strains detected in the faecal samples of commercial layer flocks in this study could pose a risk to public health, as the bacteria can spread to humans via consumption. Therefore, a paradigm for more focused biosafety measures must be considered by poultry farmers.
References
Abdel-Aziz, N. M. (2016). Detection of Salmonella species in chicken carcasses using genus specific primer belong to invA gene in Sohag city, Egypt. Veterinary World, 9(10), 1125-1128. https://doi.org/10.14202/vetworld
Andoh, L. A., Dalsgaard, A., Obiri-Danso, K., Newman, M. J., Barco, L., & Olsen, J. E. (2016). Prevalence and antimicrobial resistance of Salmonella serovars isolated from poultry in Ghana. Epidemiology & Infection, 144(15), 3288-3299. https://doi.org/10.1017/S0950268816001126
Arnam, J. S., McMurry, J. L., Kihara, M., & Macnah, R. A. (2004). Analysis of an engineered Salmonella flagellar fusion protein, FliR-FlhB. Journal of Bacteriology, 186(8), 2495-2498. ..https://doi.org/10.1128/JB.186.8
Bangera, S. R., Umakanth, S., Chowdhury, G., Saha, R. N., Mukhopadhyay, A. K., & Ballal, M. (2019). Poultry: a receptacle for non-typhoidal Salmonellae and antimicrobial resistance. Iranian Journal of Microbiology, 11(1), 31-38. https://pubmed.ncbi.nlm.nih.gov/30996829/
Carver, T., Harris, S. R., Berriman, M., Parkhill, J., & McQuillan, J. A. (2012). Artemis: an integrated platform for visualization and analysis of high-throughput sequence-based experimental data. Bioinformatics, 28(4), 464-469. https://doi.org/10.1093/bioinformatics/btr703
CLSI. (2020). Performance Standards for Antimicrobial Susceptibility Testing. 30th Ed. Clinical and Laboratory Standards Institute (CLSI).
da Silva, F. B., Ferreira, M. R. A., Waller, S. B., Sobrinho, I. D. J., Dias, M., de Freiras, S. B., Hartwig, D. D., Conceição, F. R., Stella, A. E., & Moreira, C. N. (2024). Multidrug-resistant non-typhoidal Salmonella in seasoned chicken meat. Food Control, 160, 110347. https://doi.org/10.1016/j.foodcont.2024.110347
Diab, M. S., Thabet, A. S., Elsalam, M. A., Ewida, R. M., & Sotohy, S. A. (2023). Detection of virulence and β-lactamase resistance genes of non-typhoidal Salmonella isolates from human and animal origin in Egypt ‘one health concern’. Gut Pathogens, 15(1), 16. doi: 10.1186/s13099-023-00542-3
Eguale, T. (2018). Non-typhoidal Salmonella serovars in poultry farms in central Ethiopia: Prevalence and anti-microbial resistance. BMC Veterinary Research, 14, 217. https://doi.org/10.1186/s12917-018-1539-4
EUCAST. (2021). The European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. http://www.eucast.org
Fagbamila, I. O., Barco, L., Mancin, M., Kwaga, J., Ngulukun, S. S., Zavagnin, P., Lettini, A. A., Lorenzetto, M., Abdu, P. A., Kabir, J., Umoh, J., Ricci, A., & Muhammad, M. (2017). Salmonella serovars and their distribution in Nigerian commercial chicken layer farms. PLoS One, 12(3), e0173097. https://doi.org/10.1371/journal.pone.0173097
Fagbamila, I. O., Ramon, E., Lettini, A. A., Muhammad, M., Longo, A., Antonello, K., Aworh, M. K., Kwaga, J. K. P., Abdu, P. A., Umoh, J. U., Kabir, J. A., Ricci, A., & Barco, L. (2023). Assessing the mechanisms of multi-drug resistant non-typhoidal Salmonella (NTS) serovars isolated from layer chicken farms in Nigeria. PLoS One, 18(9), e0290754. https://doi.org/10.1371/journal.pone.0290754
Fashae, K., Ogunsola, F. T., Aarestrup, F. M., & Hendriksen, R. S. (2010). Antimicrobial susceptibility and serovars of Salmonella from chickens and humans in Ibadan, Nigeria. The Journal of Infection in Developing Countries, 4(8), 484-494. https://doi.org/10.3855/jidc.909
Feltha, R., & Barrow, G. (2003). Cowan and steel’s: manual for the identification of medical bacteria. 3rd ed. Cambridge. Cambridge University Press. pp. 353.
Hendriksen, J. J., Lee, H. J., Bradshaw, A. J., Namba, K., Chevance, F. F. V., Minamino, T., & Hughes, K. T. (2021). Genetic analysis of the Salmonella FliE protein that forms the base of the flagellar axial structure. mBio, 12, 10.1128/mbio.02392-21. https://doi.org/10.1128/mbio.02392-21
Hensel, M., Hinsley, A. P., Nikolaus, T., Sawers, G., & Berks, B. C. (1999). The genetic basis of tetrathionate respiration in Salmonella typhimurium. Molecular Microbiology, 32(2), 275-287. https://doi.org/10.1046/j.1365-2958.1999. 01345.x
Hinsley, A. P., & Berks, B. C. (2002). Specificity of respiratory pathways involved in the reduction of sulfur compounds by Salmonella enterica. Microbiology, 148(11), 3631-3638. https://doi.org/10.1099/00221287-148-11-3631
Huoy, L., Nasirzadeh, L., Phan, K., Tieng, S., Sternberg-Lewerin, S., & Bongcam-Rudloff, E. (2025). Antimicrobial resistance and serotype distribution of Salmonella spp. isolated from fresh foods in Cambodia. Journal of Applied Microbiology, 136(6), lxaf137. https://doi.org/10.1093/jambio/lxaf137
Im, M. C., Jeong, S. J., Kwon, Y., Jeong, O., Kang, M., & Lee, Y. J. (2015). Prevalence and characteristics of Salmonella spp. isolated from commercial layer farms in Korea. Poultry Science, 94(7), 1691-1698. https://doi.org/10.3382/ps/pev137
Kuhlen, L., Abrusci, P., Johnson, S., Gault, J., Deme, J., Caesar, J., Dietsche, T., Mebrhatu, M. J., Ganief, T., Macek, B., Wagner, S., Robinson, C. V., & Lea, S. M. (2018). Structure of the core of the type III secretion system export apparatus. Nature Structural & Molecular Biology, 25, 583-590. https://doi.org/10.1038/s41594-018-0086-9
Lettini, A. A., Than, V. T., Marafin, E., Longo, A., Antonello, K., Zavagnin, P., Barco, L., Mancin, M., Cibin, V., Morini, M., Sao, D. T., Nguyen, T., Pham, T. H., Le, L., Nguyen, D. I., & Ricci, A. (2016). Distribution of Salmonella serovars and antimicrobial susceptibility from poultry and swine farms in central Vietnam. Zoonoses and Public Health, 63(7), 569-576. https://doi.org/10.1111/zph.12265
Magiorakos, A. P., Srinivasan, A., Carey, R. P., Carmeli, Y., Falagas, M. E., Giske, C. G., Harbarth, S., Hindler, J. F., Kahlmeter, G., Olssen-Liljequist, B., Paterson, D. L., Rice, L. B., Stelling, J., Struelens, M. J., Vatopoulos, A., Weber, J. T., & Monnet, D. L. (2012). Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clinical Microbiology and Infection, 18, 268-281. https://doi.org/10.1111/j.1469-0691.2011.03570
Majowicz, S. E., Musto, J., Scallan, E., Angulo, F. J., Kirk, M., O’Brien, S. J., Jones, T. F., Fazil, A., & Hoekstra, R. M. (2010). The global burden of non-typhoidal Salmonella gastroenteritis. Clinical Infectious Diseases, 50(6), 882-889. https://doi.org/10.1086/650733
Malorny, B., Hoorfar, J., Bunge, C., & Helmuth, R. (2003). Multicenter validation of the analytical accuracy of Salmonella PCR: towards an international standard. Applied and Environmental Microbiology, 69(1), 290-296. https://doi.org/10.1128/AEM.69.1.290-296.2003
Mihaiu, L., Lapusan, A., Tanasuica, R., Sobolu, R., Mihaiu, R., Oniga, O., & Mihaiu M. (2014). First study of Salmonella in meat in Romania. The Journal of Infection in Developing Countries, 8(1), 50-58. https://doi.org/10.3855/jidc.3715
Mohammed, B. T. (2022). Identification and bioinformatic analysis of invA gene of Salmonella in free range chicken. Brazilian Journal of Biology, 84, e263363. https://doi.org/10.1590/1519-6984.263363
Neelawala, R. N., Edison, L. K., & Kariyawasam, S. (2024). Pre-harvest non-typhoidal Salmonella control strategies in commercial layer chickens. Animals, 14(24), 3578. https://doi.org/10.3390/ani14243578
O’Brien, S. J. (2013). The “Decline and Fall” of non typhoidal Salmonella in the United Kingdom. Clinical Infectious Diseases, 56(5), 705-710. https://doi.org/10.1093/cid/cis967
Pande, V. V., Gole, V. C., McWhorter, A. R., Abraham, S., & Chousalkar, K. K. (2015). Antimicrobial resistance of non-typhoidal Salmonella isolates from egg layer flocks and egg shells. International Journal of Food Microbiology, 203, 23-26. https://doi.org/10.1016/j.ijfoodmicro.2015.02.025
Park, J. H., Kim, H. S., Yim, J. H., Kim, Y. J., Kim, D. H., Chon, J. W., Kim, H., Om, A. S., & Seo, K. H. (2017). Comparison of the isolation rates and characteristics of Salmonella isolated from antibiotic-free and conventional chicken meat samples. Poultry Science, 96, 2831-2838. https://doi.org/10.3382/ps/pex055
Pui, C. F., Wong, W. C., Chai, L. C., Tunung, R., Jeyaletchumi, P., Noor Hidayah, M. S., Ubong, A., Farinazleen, M. G., Cheah, Y. K., & Son, R. (2011). Salmonella: A foodborne pathogen. International Food Research Journal, 18, 465-473. https://doi.org/10.3382/ps/pex055
Raufu, I. A., Ahmed, O. A., Aremu, A., Ameh, J. A., Timme, R. E., Hendriksen, R. S., & Ambali, A. (2021). Antimicrobial and genomic characterization of Salmonella Nigeria from pigs and poultry in Ilorin, North-central, Nigeria. The Journal of Infection in Developing Countries, 15(12), 1899-1909. https://doi.org/10.3855/jidc.15025
Salem, M., Ghabbour, R., Mostafa, N. F., Awad, A., & Younis, G. (2026). Molecular characterization of Salmonella isolates from poultry in Egypt: Detection of virulence, ESBLs genes and antimicrobial resistance profiling. Veterinary Research Communications, 50(1), 44. doi: 10.1007/s11259-025-10936-0
Steele-Mortimer, O., Brumell, J. H., Knodler, L. A., Meresse, S., Lopez, A., & Finlay, B. B. (2002). The invasion associated type III secretion system of Salmonella enterica serovar Typhimurium is necessary for intracellular proliferation and vacuole biogenesis in epithelial cell. Cellular Microbiology, 4(1), 43-54. https://doi.org/10.1046/j.1462-5822.2002. 00170.x
Stoffels, L., Krehenbrink, M., Berks, B. C., & Unden, G. (2012). Thiosulfate reduction in Salmonella enterica is driven by the proton motive force. Journal of Bacteriology, 194(2), 475-485. https://doi.org/10.1128/JB.06014-11
World Health Organization. (2019). Critically important antimicrobials for human medicine 6th Revision. Ranking of medically important antimicrobials for risk management of antimicrobial resistance due to non-human use. https://www.who.int/publications/i/item/9789241515528
Yanestria, S. M., Rahmaniar, R. P., Wibisono, F. J., & Effendi, M. H. (2019). Detection of invA gene of Salmonella from milkfish (Chanos chanos) at Sidoarjo wet fish market, Indonesia, using polymerase chain reaction technique. Veterinary World, 12(1), 170-175. https://doi.org/10.14202/vetworld.2019.170-175
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Gbenga Adedeji Adewumi, Abiodun Tola Seriki, Felix Oluwasegun Alao, Cynthia Jesutomisin Komolafe, Faith Temitope Oloyede

This work is licensed under a Creative Commons Attribution 4.0 International License.




