SYNTHESIS, CHARACTERIZATIONS AND CORROSION INHIBITION PERFORMANCE OF TRANSITION METAL (II) COMPLEXES DERIVED FROM; ACETOPHENONE AND 2,4-DINITROPHENYLHYDRAZINE
DOI:
https://doi.org/10.33003/fjs-2026-1003-4794Keywords:
Hydrazone ligand, Metal(II) complexes, Corrosion inhibition, FTIR spectroscopy, PhysisorptionAbstract
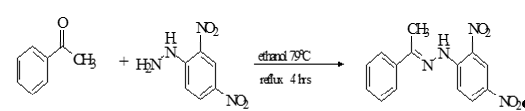
This study reports the synthesis, characterization, and corrosion inhibition performance of a hydrazone ligand derived from acetophenone and 2,4-dinitrophenylhydrazine, along with its Zn(II), Cu(II), Ni(II), Co(II), and Mn(II) complexes. The ligand and complexes were synthesized via condensation and metal-chelation reactions, and characterized using melting point determination, solubility tests, FTIR spectroscopy, conductivity measurements, and magnetic susceptibility analysis. Corrosion inhibition on mild steel was evaluated using the weight-loss method in 0.1 M HCl at varying concentrations (0.2–0.6 g/L) and temperatures (303–323 K). Results confirmed successful formation of the ligand (67.5% yield) and complexes (60.37–75.24% yield). The ligand melted at 120°C, while complexes decomposed between 160–175°C. Color changes from yellow (ligand) to pale yellow [Co(II)], brown [Cu(II)], yellow [Ni(II), Zn(II)], and black [Mn(II)] confirmed complexation. All compounds were soluble in polar solvents but insoluble in non-polar media. Conductivity measurements (3.57–8.47 Ω⁻¹cm²mol⁻¹) confirmed non-electrolytic behavior with coordinated chloride ions. Magnetic susceptibility data revealed high-spin octahedral geometries for Co(II) (5.01 B.M.), Ni(II) (2.80 B.M.), Mn(II) (7.00 B.M.), and distorted octahedral for Cu(II) (2.10 B.M.), while Zn(II) was diamagnetic. FTIR analysis confirmed coordination through azomethine nitrogen (C=N shift from 1618 cm⁻¹ to 1566–1600 cm⁻¹) and N–H groups, with new M–N bands at 732–899 cm⁻¹. Corrosion inhibition efficiency increased with concentration for all inhibitors. At 0.6 g/L and 303 K, efficiency followed: Zn(II) (85.19%) > Cu(II) (75.18%) > Ni(II) (68.83%) > Co(II) ≈ Mn(II) (67.13%) > Ligand (66.37%), with corrosion rates significantly reduced compared to the blank (249.22 mmy⁻¹).
References
Abdallah, M., El-Dafrawy, S. M., Sobhi, M., & Elwahy, A. H. M. (2020). Corrosion inhibition of mild steel in acidic solution using hydrazone derivatives. Journal of Molecular Liquids, 304, 112723.
Bello, A., Lawal, A., & Sadiq, I. U. (2022). Synthesis, characterization and corrosion inhibition studies of hydrazone metal complexes on mild steel in acidic medium. Journal of Coordination Chemistry, 75(9), 1341–1356.
Bentiss, F., Lebrini, M., & Lagrenée, M. (2019). Thermodynamic characterization of metal dissolution and inhibitor adsorption processes in mild steel corrosion. Corrosion Science, 51, 2165–2173.
El-Ashry, E. S. H., Kassem, A. A., & Abd El-Haleem, S. M. (2019). Hydrazone derivatives as corrosion inhibitors for steel in acidic media. Materials Chemistry and Physics, 233, 121–130.
Fontana, M. G. (2005). Corrosion Engineering (3rd ed.). McGraw-Hill, New York.
Muhammad, S., Abdullahi, A., and Bello, I. (2021). Corrosion Inhibition studies of organiccompounds on mild stell in acidic medium. FUDMA Journal of Science, 5(2), 120-128.
Nakamoto, K. (2009). Infrared and Raman Spectra of Inorganic and Coordination Compounds (6th ed.). John Wiley & Sons, New Jersey.
Obot, I. B., & Ebenso, E. E. (2020). Organic corrosion inhibitors for mild steel in acidic environments: A review. International Journal of Electrochemical Science, 15, 1–28. https://doi.org/10.1002/9780470405888
Revie, R. W., & Uhlig, H. H. (2008). Corrosion and Corrosion Control (4th ed.). John Wiley & Sons, New York. https://doi.org/10.1002/9780470277270
Singh, A., Ahamad, I., & Quraishi, M. A. (2021). Hydrazone derivatives as corrosion inhibitors for mild steel in hydrochloric acid. Corrosion Science, 53, 247–254. https://doi.org/10.1016/j.corsci.2020.108915
Umoren, S. A., Solomon, M. M., & Udosoro, I. I. (2022). Effect of temperature and concentration on corrosion inhibition of mild steel using organic inhibitors. Journal of Adhesion Science and Technology, 36, 1421–1440. https://doi.org/10.1080/01694243.2021.1988765
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Halliru Muhammad, Ishaq Yahaya Lawan, Aminu Ahmad, Sunusi Yahaya, Khadija Aminu Mahmoud

This work is licensed under a Creative Commons Attribution 4.0 International License.




