ACUTE AND SUB-ACUTE ORAL TOXICITY EVALUATION OF METHANOL LEAF EXTRACT OF Vitellaria Paradoxa gaertn F. IN WISTAR RATS
DOI:
https://doi.org/10.33003/fjs-2026-1003-4729Keywords:
Acute toxicity, Sub-acute toxicity, Vitellaria paradoxa methanol leaf extractAbstract
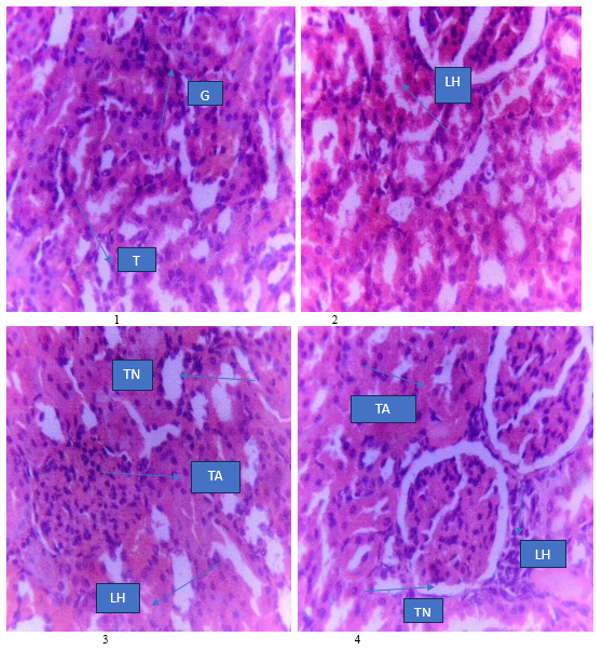
Toxicity studies of medicinal plants are essential for regulatory approval and for their therapeutic application Aim:The study aimed at evaluating the acute and sub-acute toxicity of the methanol leaf extract of Vitellaria paradoxa Gaertn F. in Wistar rats.Methods:Phytochemical screening of the extract was done using the methods of Trease and Evans (2004).The LD50 was determined using OECD (2008) Guideline 425, while the sub-acute toxicity test was conducted for 14 days at extract doses of 250, 500 and 1000 mg/kg (n=10) using OECD(2008)Guideline407.Carbohydrates,alkaloids,flavonoids,saponins,tannins, steroids/terpenes and cardiac glycosides were found in the extract but anthraquinones were absent. No signs and symptoms of toxicity, and no mortality were recorded in the acute toxicity test and the LD50 of the extract was estimated to be greater than 5000 mg/kg. Sub-acute toxicity test of the extract produced significant increases (p<0.05) in liver enzymes such as ALT and AST at extract doses tested. All the extract doses significantly decreased (p<0.05) serum urea levels, but serum creatinine levels, potassium and chloride ions concentrations were similar to those of the control group. Histological examination of the liver and kidneys showed that the extract produced slight hepatic necrosis and tubular necrosis respectively. All extract doses tested did not significantly (p>0.05) alter haematological parameters in Wistar rats.The methanol leaf extract of Vitellaria paradoxa is non-toxic in acute toxicity test, but it should be used with caution on prolonged administration as it could be toxic to liver and kidneys.
References
Adeyemi, O.T., Adeyemi, O.S. and Oguntoye, S.O. (2022). Phytochemical and pharmacological studies of some Nigerian medicinal plants. Journal of Ethnopharmacology, 285:114-128
Arsad, S.S., Esa, N.M., Hamzah, H., Othman, F. (2013). Evaluation of acute, sub-acute and sub-chronic oral toxicity of Raphidophora decursiva (Roxb.) Schott extract in male Sprague Dawley rats. Journal of Medicinal Plant Research, 7(4): 3030-3040.
Bamigbade, A. A., Olaleye, M. T., & Adewusi, E. A. (2018). Therapeutic potential of shea butter: Phytochemistry and pharmacological perspectives. Phytomedicine, 49, 152-166.
Bancroft, J.D., and Gamble, M. (2008). Haematoxylin and Eosin staining methods. In: Bancroft, J.D and Gamble, M. (Eds) Theory and Practice of Histological Techniques, Churchill Livingstone, pages 126-138.
Barnes, P.M., Bloom, B., and Nahin, R. (2008). Complementary and Alternative Medicine Use among Adults and Children in United States. CDC National Health Statistics Report No 12.
Burtis, C. A., Ashwood, E. R., and Bruns, D. E. (2017). Tietz Textbook of Clinical Chemistry and Molecular Diagnostics (6th ed.). Elsevier.
Chung, K. T., Wong, T. Y., Wei, C. I., Huang, Y. W., & Lin, Y. (1998). Tannins and human health: A review. Critical Reviews in Food Science and Nutrition, 38(6): 421–464.
Craig, R.G. (2007). Interactions between chronic renal disease and periodontal disease. Oral Disease, 1491): 1-7.
Diaz, G.J. (2015). Toxicosis by plant alkaloids in humans and animals in Colombia. Toxins, 7(12): 5408 – 5416 doi: 10.3390/26690479.
Ekor, M., Odetola, A.A. and Ogunmodede, O.S. (2020). Toxicological evaluation of herbal medicines in Nigeria. Frontiers in Pharmacology, 11:129–138.
Ernst, E., Schmidt, K., and Wider, B. (2005). Complementary therapies for asthma: An overview of systematic reviews. Respiratory Medicine, 99 (10): 1235-1242
Francis, G., Kerem, Z., and Makkar, H. P. S. (2002).The potential of plant saponins as immunomodulatory and anti-tumor agents. Journal of Agriculture and Food Chemistry, 50 (26):7771-7780
Hoffbrand, A. V., Moss, P. A. H., & Pettit, J. E. (2016). Essential Haematology (7th ed.). Wiley-Blackwell.
Karou, D., Dicko, M.H., and Traore, A.S. (2005).Antioxidant and antibacterial activities of Anogeisus leiocarpus (DC) Guill. and Perr. leaf extracts. African Journal of Biotechnology, 4(10):1113-1117.
McLean, B. J. (2015). Herbal toxicity: Understanding risks and benefits of plant-based medicines. Phytomedicine Journal, 22(5), 150-165.
Okaiyeto, K., and Oguntibeju, O.O. (2021). Medicinal plants and their bioactive compounds: A review of their potential health benefits. Journal of Pharmacy and Pharmacology, 73 (6):741-755.
Organisation for Economic Co-operation and Development (OECD)(2008a). OECD Guideline for the Testing of Chemicals, Section 4: Test No. 407 – Repeated Dose 28-Day Oral Toxicity Study in Rodents. OECD Publishing, Paris. https://doi.org/10.1787/9789264070684-en
Organisation for Economic Co-operation and Development (OECD)(2008b). Acute Oral Toxicity – Up and Down Procedure: OECD Guidelines for testing of chemicals, Guideline 425: 1-27.
Ogunlakin, A. O., Ogunleye, A.J., and Ojo, O.A. (2023). Assessment of hematological and biochemical effects of plant extracts in Wistar rats. Heliyon, 9(4):e14652.
Oyeleke, S. A., Ajayi, O.A., and Owoyemi, A.A.(2015). Toxicological evaluation of herbal remedies in experimental models. Nigerian Journal of Basic and Applied Sciences, 23(2), 92–98.
National Institute of Health Guidelines for the Care of Laboratory Animals (Production no 85-23, Revised, 1996)
Reed, J.D. (1995). Nutritional toxicology of tannins and related polyphenols in forage legumes. Journal of Animal Science, 73(5): 1516-1528. https://doi.org/10.2527/1995.7351516x
Salin, O.O., Olakunle, J.O., and Okunlola, A.O. (2021). Evaluation of sub-chronic toxicity of herbal extracts in experimental animals. Journal of Ethnopharmacology, 267, 113–121.
Stevens, L. A., Coresh, J., Greene, T., and Levey, A. S. (2006). Assessing kidney function—Measured and estimated glomerular filtration rate. New England Journal of Medicine, 354(23), 2473–2483.
Tanko, Y., Abubakar, A., and Mohammed A. (2013). Hematological and biochemical effects of medicinal plant extracts in rats. Journal of Applied Pharmaceutical Science, 3(1), 123–128.
Trease, G. E. and Evans, W.C. (2004) Pharmacognosy 13th Edition, Balliere, Tindall and Cansell Ltd. London
Wachtel-Galor, S., & Benzie, I. F. F. (2011). Herbal medicine: An introduction to its history, usage, regulation, current trends, and research needs. In: I. F. F. Benzie & S. Wachtel-Galor (Eds.), Herbal Medicine: Biomolecular and Clinical Aspects (2nd ed.). CRC Press/Taylor & Francis. Pages 1-10.
W.H.O (2008). Traditional Medicines. Available online: http://www.who.int/mediacentre/factsheets/fs134/en/accessed [13th February, 2026]
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 AbdulFatai Jimoh, Halima Mohammed, Rukayyat Bukola Oloyede, Musa Itopa Yakubu, Danjuma Mallam, Asma'u I-J Bashir, Micah Timothy, Safiya Suleiman Sambo

This work is licensed under a Creative Commons Attribution 4.0 International License.




