ANTIMICROBIAL RESISTANCE CARRIAGE AMONG EMERGENCY AND IN-HOSPITAL PATIENTS IN BENIN CITY, NIGERIA
DOI:
https://doi.org/10.33003/fjs-2026-1004-4598Keywords:
Antimicrobial resistance, Paediatric population, Methicillin-resistant Staphylococcus aureus, Extended-spectrum beta-lactamase, Carbapenemase-producing Enterobacterales, NigeriaAbstract
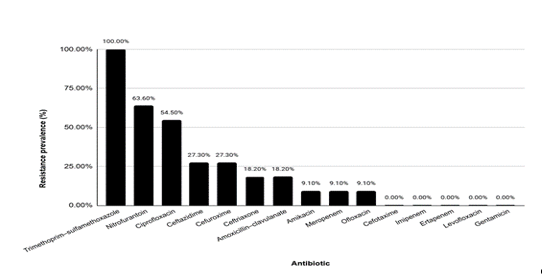
Antimicrobial resistance (AMR) is an escalating threat in sub-Saharan Africa, where hospital-based paediatric data remain limited. This study assessed the prevalence, distribution, and resistance patterns of microorganisms among children receiving care at a tertiary hospital in Benin City, Nigeria. This was a hospital-based cross-sectional analysis of paediatric patients with linked clinical and microbiological data at the University of Benin Teaching Hospital. Antimicrobial susceptibility testing was performed using the Kirby–Bauer disc diffusion method and interpreted according to Clinical and Laboratory Standards Institute criteria. Resistance phenotypes included methicillin-resistant Staphylococcus aureus (S. aureus; MRSA), extended-spectrum beta-lactamase (ESBL) production, carbapenemase production, and multidrug resistance (MDR; resistance to ≥3 antimicrobial classes). The primary outcome was any AMR at the patient level. Among 44 unique patients, AMR was detected in 12 (27.3%; 95% CI, 15.0–42.8). A total of 70 isolates were identified, dominated by S. aureus (50.0%) and Enterobacterales (25.7%). MRSA accounted for 20.0% of S. aureus isolates. Among Enterobacterales, 38.9% were ESBL-producing, and 38.9% were identified as carbapenemase-producing. Antibiotic-specific resistance was highest for trimethoprim–sulfamethoxazole (100%), nitrofurantoin (63.6%), and ciprofloxacin (54.5%), whereas phenotypic carbapenem resistance was low. AMR was strongly associated with emergency presentation (100.0% vs. 6.5% in non-emergency settings; p<0.001). In conclusion, more than one-quarter of paediatric patients in this tertiary setting had clinically significant AMR, characterised by MRSA and high ESBL prevalence among Enterobacterales. Resistance was concentrated in high-acuity hospital pathways, underscoring the need for strengthened antimicrobial stewardship, targeted infection prevention, and enhanced laboratory surveillance in tertiary paediatric care in Nigeria.
References
Adekanmbi, O., Popoola, O., Fowotade, A., Idowu, O., Ogunbosi, B., Lakoh, S., Adebiyi, I., Ayandipo, O., & Iyun, A. O. (2024). Prevalence of rectal carbapenem resistant Enterobacterales carriage among patients attending healthcare facilities in Ibadan, Nigeria: a descriptive study. BMC Infectious Diseases, 24(1), 726. https://doi.org/10.1186/s12879-024-09627-z
Adekanmbi, O., Popoola, O., Fowotade, A., Idowu, O., Ogunbosi, B., Lakoh, S., Adebiyi, I., Ayandipo, O., & Iyun, A. O. (2024). Prevalence of rectal carbapenem resistant Enterobacterales carriage among patients attending healthcare facilities in Ibadan, Nigeria: a descriptive study. BMC Infectious Diseases, 24(1), 726. https://doi.org/10.1186/s12879-024-09627-z
Bauer, A. W., Kirby, W. M., Sherris, J. C., & Turck, M. (1966). Antibiotic Susceptibility Testing by a Standardized Single Disk Method. American Journal of Clinical Pathology, 45(4_ts), 493–496. https://doi.org/10.1093/AJCP/45.4_TS.493
Centers for Disease Control and Prevention. (2019). Antibiotic resistance threats in the United States, 2019. https://www.cdc.gov/antimicrobial-resistance/media/pdfs/2019-ar-threats-report-508.pdf
Cheesbrough, M. (2006). District Laboratory Practice in Tropical Countries, Part 2. In District Laboratory Practice in Tropical Countries, Second Edition (2nd Edition). Cambridge University Press. https://doi.org/10.1017/CBO9780511543470
Clinical & Laboratory Standards Institute. (2023). Clinical & Laboratory Standards Institute: CLSI Guidelines. https://clsi.org/
Ezeh, C. K., Digwo, D. C., Okeke, I. A., Elebe, P. C., & Ezeh, E. O. (2024). A systematic review and meta-analysis on the prevalence of extended-spectrum beta-Lactamase-Producing Klebsiella Pneumonia in Nigeria. African Health Sciences, 24(3), 30–40. https://doi.org/10.4314/ahs.v24i3.5
Gahimbare, L., Muvunyi, C. M., Guessennd, N. A. K., Rutanga, J. P., Gashema, P., Fuller, W., Mwamelo, A. J., Coulibaly, S. O., Mosha, F. S., Perovic, O., Tali-Maamar, H., & Yahaya, A. A. (2024). Antimicrobial Resistance in the WHO African Region: A Systematic Literature Review 2016-2020. Antibiotics (Basel, Switzerland), 13(7). https://doi.org/10.3390/antibiotics13070659
Jewoola, O. O., Bode-Sojobi, I. O., Ogunsola, F. T., & Okonji, P. E. (2020). High carriage rates of extended-spectrum beta-lactamase-producing enterobacteriaceae in children at admission into paediatric wards of a university teaching hospital in Lagos, Nigeria. The Nigerian Postgraduate Medical Journal, 27(2), 136–142. https://doi.org/10.4103/npmj.npmj_209_19
Laxminarayan, R., Duse, A., Wattal, C., Zaidi, A. K. M., Wertheim, H. F. L., Sumpradit, N., Vlieghe, E., Hara, G. L., Gould, I. M., Goossens, H., Greko, C., So, A. D., Bigdeli, M., Tomson, G., Woodhouse, W., Ombaka, E., Peralta, A. Q., Qamar, F. N., Mir, F., … Cars, O. (2013). Antibiotic resistance—the need for global solutions. The Lancet Infectious Diseases, 13(12), 1057–1098. https://doi.org/10.1016/S1473-3099(13)70318-9
Magiorakos, A. P., Srinivasan, A., Carey, R. B., Carmeli, Y., Falagas, M. E., Giske, C. G., Harbarth, S., Hindler, J. F., Kahlmeter, G., Olsson-Liljequist, B., Paterson, D. L., Rice, L. B., Stelling, J., Struelens, M. J., Vatopoulos, A., Weber, J. T., & Monnet, D. L. (2012). Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clinical Microbiology and Infection, 18(3), 268–281. https://doi.org/10.1111/J.1469-0691.2011.03570.X
Nigeria Centre for Disease Control. (2017). Antimicrobial Use and Resistance in Nigeria: Situation Analysis and Recommendations. https://ncdc.gov.ng/themes/common/docs/protocols/56_1510840387.pdf
Nordmann, P., Naas, T., & Poirel, L. (2011). Global spread of Carbapenemase-producing Enterobacteriaceae. Emerging Infectious Diseases, 17(10), 1791–1798. https://doi.org/10.3201/eid1710.110655
O’Neil, J. (2016). Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. https://amr-review.org/sites/default/files/160525_Final%20paper_with%20cover.pdf
Paterson, D. L., & Bonomo, R. A. (2005). Extended-spectrum beta-lactamases: a clinical update. Clinical Microbiology Reviews, 18(4), 657–686. https://doi.org/10.1128/CMR.18.4.657-686.2005
Sadauki, A. H., Olorukooba, A. A., Balogun, M. S., Dalhat, M. M., Waziri, H., Abdulaziz, M. M., Umeokonkwo, C. D., Hassan-Hanga, F., & Sabitu, K. (2022). Nasal carriage of meticillin-resistant Staphylococcus aureus among children living with HIV attending Infectious Diseases Clinics in Kano, Nigeria. Infection Prevention in Practice, 4(2), 100213. https://doi.org/10.1016/j.infpip.2022.100213
Umeokonkwo, C. D., Onah, C. K., Adeke, A. S., Igwe-Okomiso, D. O., Umeokonkwo, A. A., Madubueze, U. C., Dauda, S. O., Okeke, K. C., Versporten, A., Oduyebo, O. O., Goossens, H., & Agu, A. P. (2023). Antimicrobial use among paediatric inpatients in a Nigerian tertiary hospital: A three-year point prevalence survey. Journal of Infection Prevention, 24(2), 71. https://doi.org/10.1177/17571774231152719
Williams, P. C. M., Isaacs, D., & Berkley, J. A. (2018). Antimicrobial resistance among children in sub-Saharan Africa. The Lancet Infectious Diseases, 18(2), e33–e44. https://doi.org/10.1016/S1473-3099(17)30467-X
World Health Organization. (2022). Global Antimicrobial Resistance and Use Surveillance System (GLASS) Report 2022. World Health Organization.
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Kelly Elimian, Ephraim Ibadin

This work is licensed under a Creative Commons Attribution 4.0 International License.




