COMPARATIVE ADSORPTION OF Pb²⁺ USING EDTA-MODIFIED BANANA PEEL (Musa acuminata) AND PALM KERNEL SHELL (Elaeis guineensis)
DOI:
https://doi.org/10.33003/fjs-2026-1003-4368Keywords:
Pb²⁺ adsorption, Agro-waste adsorbents, Musa acuminata, Elaeis guineensis, Surface modification, Aqueous remediationAbstract
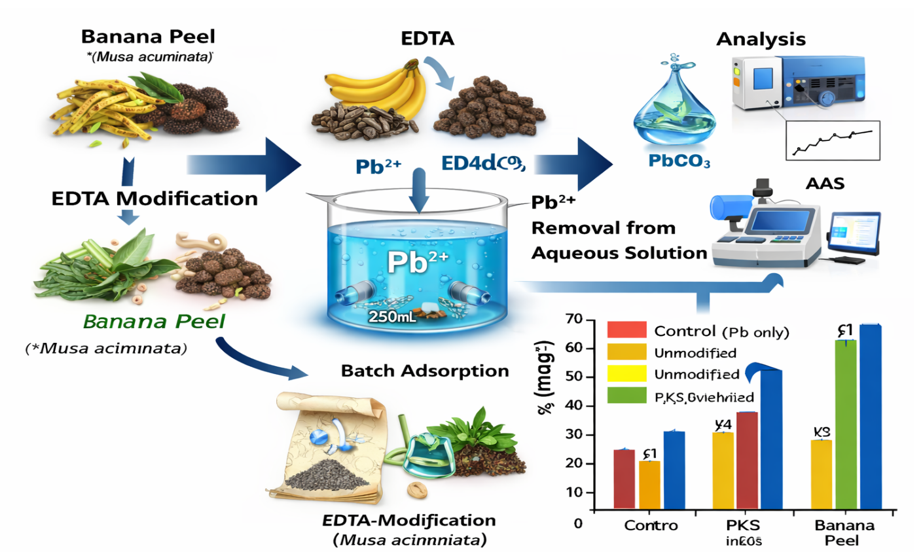
Heavy metal contamination of water resources, particularly by lead, poses serious environmental and public health risks due to its toxicity and bioaccumulation potential. In many developing regions, conventional water treatment technologies are costly and energy-intensive, necessitating affordable and sustainable alternatives. This study investigated the adsorption performance of two abundant agro-wastes in Nigeria (banana peel and palm kernel shell) as bioadsorbents for Pb²⁺ removal from aqueous solutions. The objective was to evaluate the effectiveness of raw and ethylenediaminetetraacetic acid (EDTA)-modified biomaterials and to assess the influence of surface modification on adsorption efficiency. Batch adsorption experiments were conducted using 5.0 g of adsorbent in 100 mL of 0.01 M Pb(CH₃COO)₂ solution with a contact time of 4 h under ambient conditions. Residual Pb²⁺ concentrations were determined using gravimetric precipitation as PbCO₃ and Atomic Absorption Spectroscopy (AAS). Gravimetric results showed that EDTA-modified banana peel achieved the highest Pb²⁺ removal efficiency (≈99.3%), reducing the equilibrium concentration from 1736.94 mg L⁻¹ (control) to 15.51 mg L⁻¹, followed by modified palm kernel shell with ≈97.8% removal and a residual concentration of 46.53 mg L⁻¹. Unmodified banana peel and palm kernel shell showed lower efficiencies, with equilibrium Pb²⁺ concentrations of 170.59 and 356.69 mg L⁻¹, respectively. Although high removal efficiencies were obtained, residual Pb²⁺ concentrations remained above the World Health Organization guideline value of 0.01 mg L⁻¹, indicating the need for further optimization. The study demonstrates that EDTA-modified agro-wastes are effective, low-cost bioadsorbents for Pb²⁺ remediation in aqueous systems.
References
Abel-Gawad, A. M., Shaaban, A. F., & Ebrahim, S. M. (2023). Modelling of Pb(II) biosorption kinetics using agro-waste materials. Environmental Technology & Innovation, 29, 103120.
https://doi.org/10.1016/j.eti.2022.103120
Ahmad, M. A., Ahmad, N., & Bello, O. S. (2015). Modified orange peel as adsorbent for lead(II) removal: Kinetics and equilibrium studies. Bioresource Technology, 190, 331–336.
https://doi.org/10.1016/j.biortech.2015.04.068
Ajiboye, O. T., Abioye, A. I., & Hassan, M. (2023). Adsorption behaviour of Pb(II) ions on functionalized biomass: Kinetic and isotherm studies. Environmental Nanotechnology, Monitoring & Management, 20, 100732.
https://doi.org/10.1016/j.enmm.2023.100732
Alhaji Adamu, Y. (2023). Removal of heavy metals by adasorbent produced from khaya senegalensis. FUDMA Journals of Sciences, 7(1), 29-34.
https://doi.org/10.33003/fjs-2023-0701-1168
Ali, H., Khan, E., & Sajad, M. A. (2019). Phytoremediation of heavy metals—Concepts and applications. Chemosphere, 217, 845–857.
https://doi.org/10.1016/j.chemosphere.2018.11.020
APHA. (2017). Standard methods for the examination of water and wastewater (23rd ed.). American Public Health Association.
Crini, G., & Lichtfouse, E. (2019). Advantages and disadvantages of techniques used for wastewater treatment. Environmental Chemistry Letters, 17, 145–155.
https://doi.org/10.1007/s10311-018-0785-9
Eze, C. J., Okoro, U. C., & Agbo, E. (2021). Removal of Pb(II) ions using modified groundnut shell: Equilibrium and kinetic study. SN Applied Sciences, 3(2), 210–219.
https://doi.org/10.1007/s42452-021-04210-7
Haque, M. M., Islam, R., & Hossain, M. T. (2022). Comparative evaluation of lignocellulosic biosorbents for Pb(II) ion uptake. Materials Today: Sustainability, 18, 100174.
https://doi.org/10.1016/j.mtsust.2022.100174
Haque, M. M., Rahman, M. M., & Khan, M. A. (2022). Biosorption of Pb(II) ions using modified agricultural residues. Journal of Environmental Chemical Engineering, 10(1), 106112.
https://doi.org/10.1016/j.jece.2021.106112
Li, H., Zhang, X., & Zhao, Y. (2022). Chronic kidney disease as an integrated molecular and cellular process. Frontiers in Medicine, 9, 896–905.
https://doi.org/10.3389/fmed.2022.896905
Liu, Y., Chen, Z., & Li, W. (2022). Influence of functional group modification on biosorption kinetics of Pb(II). Environmental Science and Pollution Research, 29, 13415–13425.
https://doi.org/10.1007/s11356-021-16532-9
Moyo, M., Nare, L., & Chikazaza, L. (2020). Adsorption of heavy metals from wastewater using banana peels: A review. Applied Water Science, 10(4), 142–151.
https://doi.org/10.1007/s13201-020-01242-4
Nwakogo, E. N., Okafor, V. O., & Nnaji, N. J. (2021). Surface modification of agricultural wastes for improved adsorption of Pb(II) ions. Environmental Research Communications, 3(11), 115002.
https://doi.org/10.1088/2515-7620/ac2e5a
Olayinka, K. O., Adesina, O. A., & Adejumo, A. O. (2020). EDTA-modified agro-waste biosorbents for heavy metal removal from aqueous solutions. Journal of Environmental Chemical Engineering, 8(4), 103692.
https://doi.org/10.1016/j.jece.2020.103692
Simón, F., Álvarez, C., & Martínez, A. (2022). Improvement of heavy metal removal by EDTA-functionalized biomass: A mechanistic approach. Chemosphere, 299, 134422.
https://doi.org/10.1016/j.chemosphere.2022.134422
Singh, R., & Mishra, R. (2020). Kinetic and isotherm modelling of Pb(II) adsorption onto modified lignocellulosic biomass. International Journal of Environmental Science and Technology, 17, 2123–2132.
https://doi.org/10.1007/s13762-019-02564-7
Welz, B., & Sperling, M. (2022). Atomic absorption spectrometry: Analytical chemistry by open learning (4th ed.). Wiley-VCH.
World Health Organization. (2022). Guidelines for drinking-water quality (4th ed.). WHO Press.
Wu, Y., Lin, C., & Huang, S. (2024). Functionalization of lignocellulosic wastes for enhanced heavy metal adsorption. Separation and Purification Technology, 345, 124166.
https://doi.org/10.1016/j.seppur.2024.124166
Yadav, S., & Gupta, R. (2020). Lignocellulosic biomass as biosorbents for heavy metal remediation: A review. Journal of Environmental Management, 259, 110050.
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2026 Kenneth Koomtoe Gurumyen; Babatunde Sunday Dada, Sani M. Sambo, Charity Uren Zang

This work is licensed under a Creative Commons Attribution 4.0 International License.




