PRODUCTION OF PECTINOLYTIC ENZYMES FROM BACTERIAL ISOLATES ASSOCIATED WITH OLIVE (Olea europaea) PLANTS
DOI:
https://doi.org/10.33003/fjs-2025-0912-4026Keywords:
Olive plants, Endophytic Bacteria, Orange peel powder, Pectinase production, Enzyme activityAbstract
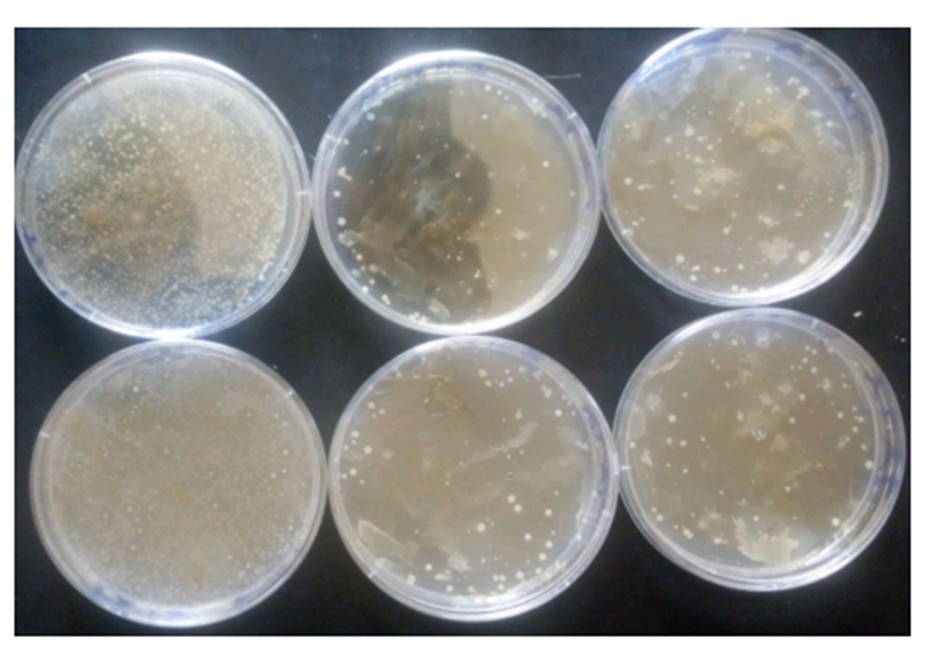
Microbial pectinases are widely used in various industries and represent a major class of industrial enzymes. Their broad applications have attracted significant research interest which include plant fiber processing, pectic waste water treatment, paper pulping, fruit juice extraction, and clarification. This study aimed to isolate endophytic bacteria from olive plants and evaluate their potential for pectinase production using the solid-state fermentation (SSF) method. Samples were collected from olive plants in a home orchard located in Zuru, Kebbi State, Nigeria. Bacterial cultures exhibiting the highest zones of inhibition were selected for enzyme production. Orange peel powder served as the pectin-rich substrate in the submerged production medium. Among the isolates, a single pectinolytic strain demonstrated enzyme activity of 1.387 U/mL. These findings suggest that endophytic bacteria associated with olive plants in Zuru have potential as industrial producers of pectinase enzymes.
References
Bouras, M., Messaoudi, B. and El Fels, L. (2019). Microorganisms isolated from Moroccan olive-mill wastes: Screening of their enzymatic activities for biotechnological use. European Scientific Journal, 15(12), 185–198. https://doi.org/10.19044/esj.2019.v15n12p185
Dhillon, G.S., Kaur, S., Brar, S.K., Verma, M. (2025). Citrus peel as a substrate for pectinase production in semi-solid fermentation.Journal of Industrial Microbiology and Biotechnology, 32(4), 147–153.
El-Tarabily, K.A. and Sivasithamparam, K. (2006). Non-streptomycete actinomycetes as biocontrol agents of soil-borne fungal plant pathogens and as plant growth promoters. Soil Biology and Biochemistry, 38, 1505–1520.
Freitas, S., Martins, L.L. and Silva, T. (2025).Thermophilic bacterial strains for industrial pectinase production.Enzyme and Microbial Technology, 38(3), 234–240.
Haile, S., Masi, C. and Tafesse, M. (2022). Isolation and characterization of pectinase-producing bacteria Serratia marcescens from avocado peel waste for juice clarification. BMC Microbiology, 22, 145. https://doi.org/10.1186/s12866-022-02536-8
Hallmann, J., Quadt-Hallmann, A., Mahaffee, W.F. and Kloepper, J.W. (1997). Bacterial endophytes in agricultural crops. Canadian Journal of Microbiology, 43(10), 895-914.
Hankin, L. and Anagnostakis, S.L. (1975). The use of solid media for detection of enzyme production by fungi. Mycologia, 67, 597–607.
Kashyap, A., Kumari, A. and Soni, S. (2022). Microbial pectinases: Classification, production, and biotechnological applications. Current Microbiology, 79, 264. https://doi.org/10.1007/s00284-022-02933-5
Miller, G.L. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry, 31(3), 426–428
Mrudula, S. and Anitharaj, R. (2025). Influence of nitrogen sources on microbial enzyme production in solid-state fermentation. African Journal of Biotechnology, 12(25), 4050–4055.
Rubinder, K., Singh, R. and Kumari, P. (2025). Characterization of pectinolytic enzymes from soil bacteria under thermophilic conditions. Biotechnology Advances, 23(2), 199–205.
Sallami, A., Rachidi, F., Lahsini, A.I., El Khedri, H., Douira, A., El Modafar, C., Medraoui, L. and Filali-Maltouf, A. (2023). Plant growth promoting (PGP) performances and diversity of bacterial species isolated from olive (Olea europaea L.) rhizosphere in arid and semi-arid regions of Morocco. Journal of Pure and Applied Microbiology, 17(4), 2165–2178. https://doi.org/10.22207/JPAM.17.4.13
Samira, R., Ali, N. and Farhad, H. (2025). Screening and identification of pectinolytic bacteria from agricultural soils. International Journal of Microbial Research, 9(1), 25–31.
Schulz, B., Boyle, C., Draeger, S., Römmert, A.K. and Krohn, K. (2002). Endophytic fungi: a source of novel biologically active secondary metabolites. Mycological Research, 106(9), 996-1004.
Shahriarinour, M., Wahab, R.A. and Salleh, A.B. (2025). Citrus pectin as a selective carbon source for screening endophytic bacteria with pectinolytic activity. Journal of Applied Sciences, 15(6), 622–628.
Strobel, G. and Daisy, B. (2023). Bioprospecting for microbial endophytes and their natural products. Microbiology and Molecular Biology Reviews, 67(4), 491–502.
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2025 Hyeladzira Bwala Yusuf, Samuel A. Emannuel, Abdulbasid Muhammad, Waye P. Jummai, Ishaku Edith, Lugard T. John, Haruna M. Nggada

This work is licensed under a Creative Commons Attribution 4.0 International License.




